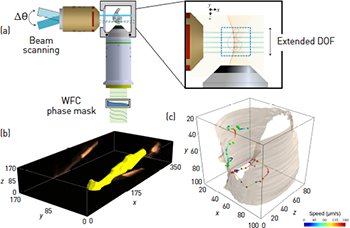
 (a) The DID-LSFM allows the production of optical sections from out-of-focus planes and enables high-resolution volumetric imaging of living samples at unprecedented speeds. Example of fast volumetric imaging using DID-LSFM: (b) 3-D rendering of a moving worm obtained with DID-LSFM after deconvolution at 8 volumes/s and (c) tracked trajectories of the cell bodies of larvae moving inside of a soil worm at 10 volumes/s. [Adapted from O. E. Olarte et al. Optica 2, 702 (2015).]
(a) The DID-LSFM allows the production of optical sections from out-of-focus planes and enables high-resolution volumetric imaging of living samples at unprecedented speeds. Example of fast volumetric imaging using DID-LSFM: (b) 3-D rendering of a moving worm obtained with DID-LSFM after deconvolution at 8 volumes/s and (c) tracked trajectories of the cell bodies of larvae moving inside of a soil worm at 10 volumes/s. [Adapted from O. E. Olarte et al. Optica 2, 702 (2015).]
Current fluorescence microscopy demands the visualization of large 3-D samples with increased sensitivity and higher resolution, and at fast imaging speed. Several imaging techniques based on point-scanning, light sheet, and pointillist strategies have been designed to tackle some of the above mentioned demands. Although successful, all of these possess an important limitation: the illuminated sections need to be carefully coupled with the detection optics in order to accomplish efficient optical sectioning. Breaking this constriction is possible, however, and by doing this provides a new degree of freedom that opens up imaginative applications for extracting information from a biological sample.
Recently, we reported an approach to decouple the illumination and detection optics in an imaging system in a passive way.1 This is based on the use of wavefront coding (WFC) combined with light sheet microscopy (LSFM).2,3 This simply involves inserting an adequate phase mask at the exit pupil of the collecting objective, extending its depth of field (DOF). As a consequence, optical sections from out-of-focus planes are possible. In addition, the light sheet can be tilted or have any desired shape, allowing inclined or engineered surfaces to be imaged. Several light sheets can also be combined, resulting in different illumination patterns for structured illumination. Finally, the light sheet can be freely and quickly moved, allowing fast volumetric imaging.
In our paper we have focused on the last case to demonstrate how the decoupled illumination-detection (DID) approach can be used to obtain high-resolution volumetric images of fast moving dynamics in living samples as well as for 3-D particle tracking at unprecedented speeds (>70 volumes/s). Since the only moving element is the light sheet, the bottleneck for acquiring faster 3-D images is no longer in the microscope itself. Our technique naturally demands better cameras (higher sensitivities, faster recording speeds), more efficient fluorescent labels and new deconvolution algorithms. Once these limitations are overcome, virtually any fast dynamic occurring in living samples will be accessible.
This work has great implications for fast volumetric imaging in several applications, including calcium dynamics, 3-D cell tracking and particle velocimetry. Furthermore, the full potential of the DID concept could be combined with 3-D lattice microscopy, standing-wave axial encoding and superresolution, and can be exploited in applications such as optogenetics and active neural stimulation.
Researchers
Omar E. Olarte, Jordi Andilla and Pablo Loza-Alvarez, ICFO—Institut de Ciencies Fotoniques, Spain
David Artigas, ICFO and Universitat Politècnica de Catalunya, Spain
References
1. O.E. Olarte et al. Optica 2, 702 (2015).
2. J. Dowski and W. T. Cathey. Appl. Opt. 34, 1859 (1995).
3. J. Huisken et al. Science 305, 1007 (2004).
