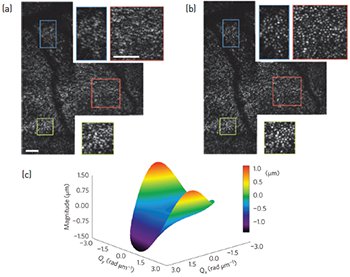
 An uncorrected photoreceptor mosaic acquired using en face OCT (a) and after computational aberration correction (b). A surface plot of a computational wavefront correction applied to an en face OCT frame (c). Scale bars = 0.25 degrees. [Reprinted by permission from Macmillan Publishers Ltd: N.D. Shemonski et al. Nat. Photon. 9, 440 (2015)]
An uncorrected photoreceptor mosaic acquired using en face OCT (a) and after computational aberration correction (b). A surface plot of a computational wavefront correction applied to an en face OCT frame (c). Scale bars = 0.25 degrees. [Reprinted by permission from Macmillan Publishers Ltd: N.D. Shemonski et al. Nat. Photon. 9, 440 (2015)]
Our eyes provide us with detailed and rich sensory information—the eyes are also a window into various biological processes. Over the past two decades, optical coherence tomography (OCT) has become the standard of care for imaging eye disease.1 However, ocular aberrations limit cellular resolution tomography of the human retina. The introduction of hardware-based adaptive optics (HAO) has enabled diffraction-limited imaging of previously unresolvable structures, such as the three-dimensional visualization of photoreceptor distributions.2 Unfortunately, HAO’s sophisticated hardware and controlling software adds considerable complexity and cost to an OCT system. This limits the number of researchers and clinicians who can benefit from HAO technology.
Computational adaptive optics (CAO) is a processing method that modifies the complex interferometric data acquired from OCT by applying a digital aberration-correction filter in the spatial frequency domain.3 This method is capable of correcting high-order optical aberrations without the use of adaptive optics hardware, such as a wavefront sensor or a deformable mirror, and has previously been demonstrated in ex vivo biological samples.
The frequent and unavoidable motion of the eye can corrupt the phase of interferometric data, preventing the use of phase-sensitive techniques such as CAO. We have developed a phase stabilization technique and automated CAO approach to computationally correct strong optical aberrations in the living human retina.4 We acquired retinal data using a standard en face OCT system operating at a 4-kHz en face line rate to overcome transverse motion. We corrected axial motion in post-processing. Next, we used an automated method to determine the optimal aberration correction filter, and then applied the filter to the computational pupil plane. Following computational aberration correction, we obtained a clearly resolved photoreceptor mosaic. These results demonstrate that CAO is applicable even under dynamic in vivo conditions.
We anticipate that CAO will supplement, or in some cases replace, HAO systems, making high-resolution imaging of retinal features widely available to researchers and clinicians.
Researchers
Nathan D. Shemonski, Fredrick A. South, Yuan-Zhi Liu, Steven G. Adie, P. Scott Carney, and Stephen A. Boppart, University of Illinois at Urbana-Champaign, USA
References
1. D. Huang et al. Science 254, 1178 (1991).
2. Y. Zhang et al. Opt. Express 14, 4380 (2006).
3. S.G. Adie et al. Proc. Natl. Acad. Sci. USA 109, 7175 (2012).
4. N.D. Shemonski et al. Nat. Photon. 9, 440 (2015).
