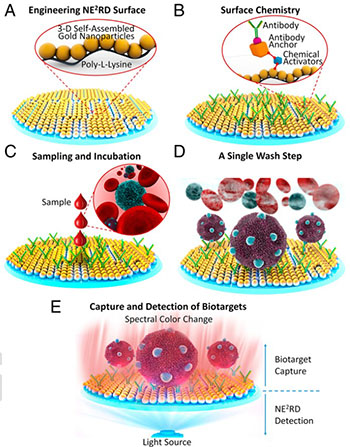
The NE2RD device consists of an engineered surface of nanospheres (A) permeated with anchored antibodies (B) that capture specific biomolecules from samples such as whole blood (C). After washing away extraneous cells and other material (D), the change in the spectral response of the plasmonic surface attributable to the biomolecules is measured (E). [Image: Yusuf Yesil/F. Inci et al., PNAS, doi: 10.1073/pnas.1510824112]
A physician, in an office or in the field, is only as good as the diagnostic data at his or her disposal; hence the drive for better, cheaper biosensors that can be leveraged at the point of care. A large team of researchers from the United States, China and Singapore now reports that it has created an integrated device that leverages plasmonics and microfluidics to overcome limitations of some previous efforts toward cheap, field-ready biosensing (PNAS, doi: 10.1073/pnas.1510824112).
The researchers believe that the device—which reportedly can detect multiple biotargets at a sensitivity far above that of conventional assays—could constitute a new platform for developing portable diagnostics in a variety of primary-care and point-of-care settings.
The quest for an integrated tool
In developing biosensing technology, the devil is in the details. An ideal technology for the point of care, particularly in under-resourced areas, would be cheap, simple, reliable, and fast, as well as capable of targeting a range of biomolecules. Many conventional biosensing techniques, however, require labeling or extensive sample preparation to smooth out variations attributable to temperature or pH changes, and can address only a limited selection of biomolecules across a narrow dynamic range.
All of that tends to drive complexity and total cost up. Recent developments in nanotechnology, and especially in plasmonics (see “Plasmonic Nanoantennas: From Nanotweezers to Plasmonic Photography,” OPN, June 2015) have addressed some, but not all, of these shortfalls. And finding a way to combine those advances into an integrated, practical device has proved a thorny problem.
Leveraging a gold nanosurface
To take that step, the multinational team, led by Ronald Davis of Stanford University, zeroed in on nanoplasmonics and microfluidics as key enabling technologies. The new tool, which the scientists have dubbed a nanoplasmonic electrical-field-enhanced resonating device—with the somewhat jocular acronym NE2RD—consists of a polystyrene (poly-L-lysine) surface, decorated with a layer of 3-D self-assembled nanoparticles 10 nm in diameter. Using surface chemistry techniques, the scientists then anchored antibodies, selected for their ability to bind to specific biomolecules of interest, to the nanosurface.
Then, samples of whole blood, serum and saliva were applied, without pre-processing, to the plasmonic surface, where the anchored antibodies grabbed onto the biomolecule of interest. The team assessed the nanoplasmonic response to the captured biotargets by measuring changes in spectra off of the surface owing to the biomolecules, using Fourier analysis.
“Unprecedented multitarget capability”
The result: NE2RD was able to detect an array of biomolecules, ranging from protein biomarkers to drugs and allergens to specific viruses—an “unprecedented multitarget capability,” in the words of the study—from a variety of different bodily fluids. The results were robust across variations in temperature, pH, and ionic strength. And the sensitivity was, in the case of one molecule (IFN-γ), three orders of magnitude better than the detection limit of the most common conventional assay.
In addition, one of the prototypes successfully integrated the NE2RD nanoplasmonic surface into a disposable, fluidic chip, pointing to its potential use in miniaturized, portable devices in a field setting. “In the future,” the research team concludes, “the current system could be integrated seamlessly with mobile platforms . . . thus enabling measurements without the need for skilled personnel.”
