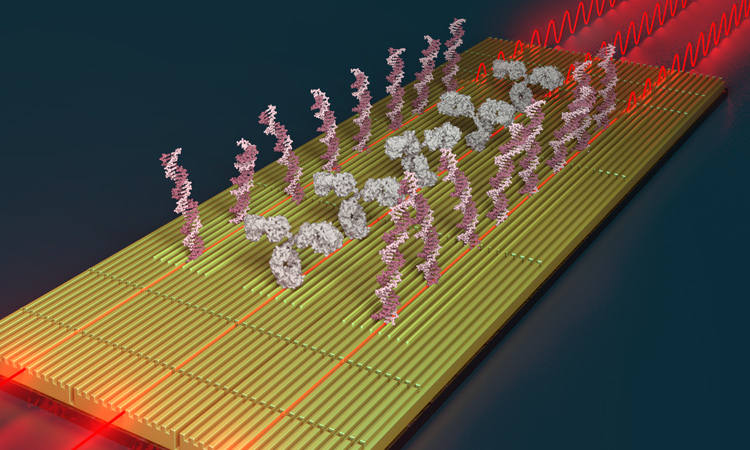
 Nanophotonic interferometric biosensor for multiplexed analysis of protein and nucleic acid biomarkers. [Dámaso Torres/ICN2]
Nanophotonic interferometric biosensor for multiplexed analysis of protein and nucleic acid biomarkers. [Dámaso Torres/ICN2]
We live amid the rise of precision medicine. Every day seems to bring news of the discovery and implementation of novel treatments to eradicate diseases, and personalized therapies to attack malignancies with impressive results. Scientists around the world are on a quest for the key remedies to reduce the burdens of cancer and of cardiac, degenerative or autoimmune disorders. But first things first: To fight a disease, first we need to find it.
Photonic biosensors exploit light’s unique properties to realize some of the more sensitive, robust and reliable sensing platforms currently available.
Early, accurate and informative diagnosis is essential for selecting the most appropriate therapy and administering it in time. Rapid identification of an infectious outbreak is crucial to stem an epidemic; early-stage detection of cancer holds extreme importance for treatment response and patient survival. Clinical diagnosis thus must evolve even more to incorporate new methods and technologies that enable rapid, simple screening of the population, to detect diseases before their physical onset.
These new diagnostic techniques will need to identify remote biological disorders that might carry pathological consequences—providing information about the disease stage and severity, and the most promising drug or treatment candidates. And, ideally, diagnostic systems should be small, portable devices that can be employed directly by those who need it: physicians, pharmacists or the patients themselves.
Photonic biosensors: SPR and beyond
One of biomedicine’s best assets in this endeavor is biosensors. These are self-integrated devices that use specific biorecognition elements—such as antibodies, DNA strands or enzymes—to capture and detect a target molecule, or analyte, present in a sample. This interaction produces a variation in an electrical, optical or mechanical property of the transducer that can be read and directly related to the amount of analyte.
Photonic biosensors exploit light’s unique properties to realize some of the more sensitive, robust and reliable sensing platforms currently available. The paramount example is the surface plasmon resonance (SPR) biosensor. This system, based on an optical phenomenon discovered between 1902 and 1912, was first sold as an analytical technology in 1990. After 30 years, SPR biosensors have emerged as routine analysis platforms in many research and pharmaceutical laboratories, commercialized worldwide. Importantly, they have served as a solid foundation for the investigation and development of new nanophotonic technologies that move beyond existing biosensors and expand their capabilities.
Yet, despite the impressive ability for nanoscale control and the vast knowledge and expertise in bioanalysis that have arisen in recent years, the optimum nanophotonic biosensor for point-of-care clinical diagnosis has remained elusive. Where do the limitations lie—and why have optical biosensors been slow to make their way into the clinic?
In this feature, we overview some of the latest advances in nanophotonic technologies for biosensing, the main challenges and limitations, and current prospects. We will also look at some promising applications of nanophotonic sensors in biomedicine, highlighting their unique capabilities and their potential to revolutionize clinical diagnosis and general healthcare in the near future.
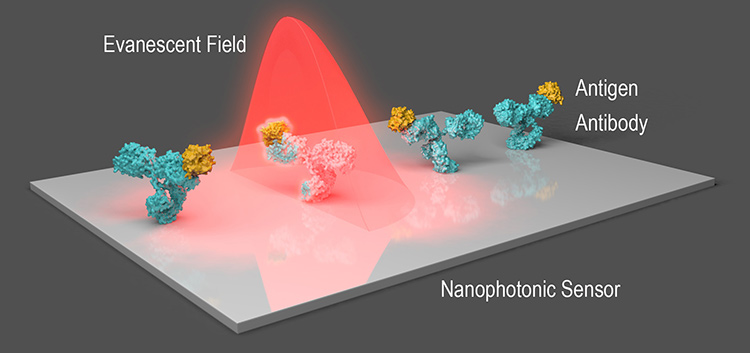 Evanescent-field sensing: In a typical nanophotonic biosensor, nanostructures on the sensor strongly enhance the incident light field; changes in refractive index at the interface due to biorecognition events (for example, antibody–antigen interactions) are read from the enhanced evanescent field. [Courtesy of the authors]
Evanescent-field sensing: In a typical nanophotonic biosensor, nanostructures on the sensor strongly enhance the incident light field; changes in refractive index at the interface due to biorecognition events (for example, antibody–antigen interactions) are read from the enhanced evanescent field. [Courtesy of the authors]
The evanescent field and its sensitivity
Most common optical biosensors are based on the so-called evanescent-field principle, generally using nanoplasmonics and silicon-photonics nanotechnology. Evanescent-field biosensors can detect biomolecular interactions occurring at the sensor surface, in real time and without labels or dyes, thereby offering a rapid, simple and noninvasive technique for studying biochemical processes or quantifying analytes.
The objective of point-of-care diagnostic biosensors is fully operational, portable devices that can be readily employed by end-users.
Plasmonic biosensors employ nanometer-sized metallic films or nanostructures (mainly gold), which absorb incident electromagnetic radiation and excite coherent electron resonances (surface plasmon polaritons) at the surface. This resonance can either propagate along the thin film (propagating SPR) or be confined to the vicinity of the nanoparticle (localized SPR, or LSPR). Either way, it generates a strong near-field enhancement that penetrates a few nanometers (10–500 nm) into the surroundings.
This evanescent field is highly sensitive to refractive-index variations in the medium. Thus, a change of composition or mass at the metal–dielectric interface translates directly to a variation of the intensity, wavelength, angle or phase of the light. The real-time interrogation of such variations during a biorecognition event (for example, an antibody–antigen interaction) provides quantitative information about both the molecule’s concentration and the affinity and kinetic parameters of the biomolecular interaction.
 A nanoplasmonic sensor: Example of a nanoplasmonic biosensor that uses a surface dotted with gold nanodisks. [Courtesy of the authors]
A nanoplasmonic sensor: Example of a nanoplasmonic biosensor that uses a surface dotted with gold nanodisks. [Courtesy of the authors]
The detection limit for nanoplasmonic biosensors ranges from 10–5 to 10–7 refractive-index units (RIU), equivalent to detection at the nanomolar (nM) level for biomolecular analysis. Demonstrations in recent years have highlighted the great analytical performance of these biosensors, with an exponential increase of publications and patents of new sensing systems for disease diagnostics. These systems can detect proteins, nucleic acids and pathogens in a few minutes, without the need of fluorescent or colorimetric tags—and with high accuracy.
From plasmonics to silicon photonics
For detecting very low amounts of biomarkers, however, the sensitivity of nanoplasmonic biosensors can be limited—or they can require light-coupling schemes that complicate their integration and miniaturization. In these cases, silicon-nanophotonics technology may offer a powerful alternative. Biosensors based on silicon resonators or waveguides also work through an evanescent-field principle, and in some cases their sensitivity can outperform that of their nanoplasmonic counterparts by more than two orders of magnitude.
In particular, interferometric sensors today constitute the most sensitive technology available, with detection limits of up to 10–8 RIU. These sensors consist of a waveguide that is partly exposed to a sensing area and partly used as reference. The evanescent field generated along the waveguide probes the refractive-index changes occurring at the sensing area, producing a difference in the phase of the light. When recombined, the interferometric signal obtained can be correlated with the analyte concentration, as with nanoplasmonic sensors.
 The bimodal waveguide (BiMW) technology developed by the Nanobiosensors and Bioanalytical Applications Group in Barcelona is one example of an interferometric biosensor. [Olalla Calvo-Lozano]
The bimodal waveguide (BiMW) technology developed by the Nanobiosensors and Bioanalytical Applications Group in Barcelona is one example of an interferometric biosensor. [Olalla Calvo-Lozano]
Interferometric biosensors have also been applied in clinical diagnostics, showing sensitivities for biomolecular markers in the picomolar to attomolar (pM–aM) range without any labelling or amplification steps. Moreover, the use of silicon technology implies an intrinsic potential for producing photonic integrated circuits (PICs) for possible incorporation into miniaturized lab-on-a-chip (LOC) sensing systems.
At the level of the lab, evanescent-field-based biosensors have shown the analytical and technological capabilities to become the next-generation diagnostic systems—they have demonstrated excellent performance and versatility, achieved outstanding detection limits, and shown promise for label-free, real-time analysis of all types of clinical biomarkers. Clinical settings, though, demand “black box” devices that can operate in point-of-care settings with the same accuracy and reliability as in the lab. It’s a significant challenge—how do we get there?
Engineering compact, ready-to-use biosensors
In one sense, the answer is simple: Miniaturize everything—but also automate it, simplify it and make it clinically reliable. But, as always, the devil is in the details.
The objective of point-of-care diagnostic biosensors is fully operational, portable devices that can be readily employed by end-users. Biosensors in clinical settings must analyze human samples like blood or urine directly, without pretreatment, providing unambiguous responses about the levels of specific molecules with an exquisite accuracy, selectivity and reproducibility. Multiplexed assays—the simultaneous detection of different markers from the same sample—are also a requirement for rapid, truly informative diagnostics.
To meet these demands, research in nanophotonic biosensors has expanded to include integration with aspects of the systems that contain them.
Advanced microfluidic systems
Microfluidics engineering has evolved tremendously over the last decade, in part thanks to progress in micro- and nanofabrication techniques that enable production of miniaturized polymer cartridges providing full and automated control of fluid transport. The latest microfluidic systems include hydraulic or pneumatic valves that can control multiple channels; microreactors; integrated pumps and waste reservoirs; and other elements.
Toward personalized therapies
The design of novel diagnostics based on nanophotonic sensing technology is opening new avenues for personalized medicine. Optical label-free biosensors for direct, real-time analysis of biomolecular interactions can be further exploited in biomedical research to boost the development of precise and individualized therapies for serious diseases, like cancer or autoimmune disorders, and to facilitate the widespread administration of such personalized approaches.
Recent efforts have started to address this complex but far-reaching goal. Nanophotonic biosensors have been already described for the label-free analysis of cell regulation pathways, as well as for so-called epigenetic alterations, such as DNA methylation or alternative splicing events. Such events have been suggested as potential targets for the earliest possible detection of tumor-initiating processes as well as for precision therapeutics. Direct, label-free analysis of such genomic disorders in pre-tumor cells affords an opportunity for developing population-screening techniques with diagnostic, prognostic and therapeutic information outputs that could substantially reduce the cancer burden.
Likewise, optical label-free biosensors have been reported as potential alternatives for live-cell analysis, which could facilitate implementation of novel personalized cancer treatments. Recently discovered cell-based immunotherapies, like the CAR-T cell therapy, might greatly benefit from devices able to achieve single-cell sensitivities and to monitor, in real time, the activity of bioengineered immune cells without labelling or staining. Such nanophotonic biosensors, if realized, could help bring down currently exorbitant treatment costs and speed up the administration of powerful immunotherapies to cancer patients worldwide.
These LOC devices can be automated and remotely controlled by software, which dramatically minimizes sample handling and manipulation by the user and scales down the biosensor system’s overall footprint. Other strategies for on-chip fluid manipulation take advantage of capillary action and material’s hydrophilicity; centrifugal forces (as in the case of compact-disc microfluidics); or even electrochemistry (in digital microfluidic systems). The latter is especially interesting, since it enables fluid transportation and mixing using nanoliter-volume droplets, which dramatically reduces the sample volume required for the analysis.
Integration in handheld devices
Current research also focuses on the integration of all optical components into miniaturized platforms, and their transfer from laboratory optical tables to small, portable devices. Replacing lasers, microscopes and spectrometers by LEDs and CMOS detectors can allow compact devices with reasonable performance to be realized.
Here, optical-biosensor development has exploited the breakthrough of extremely powerful smartphones. Nanoplasmonic sensors have been adapted to employ the flash LED lights and the high-quality cameras of commercial smartphones for operation as simple external accessories. Although these devices can’t boast the analytical sensitivity of conventional laboratory platforms, they offer the promise of truly portable point-of-care diagnostic systems.
Solving and simplifying in-coupling of light for these compact platforms, and providing high-throughput readouts from them, remain ongoing challenges. Imaging 2D microarrays at relatively large scales often results in crosstalk issues or low signal-to-noise ratios. In waveguide-based biosensors, for example, coupling the light from macrometric sources to multiple submicron waveguides in parallel is an arduous task.
To get past these problems, new nanophotonic designs incorporating gratings, photonic crystals or integrated tapers, which may offer an efficient and cost-effective solution, should be investigated and implemented in the sensor fabrication process. Also worth researching could be data processing and machine learning, to enhance the acquisition and use of optical signals by boosting the signal-to-noise ratios and minimizing crosstalk for multiplexed assays, thereby providing fast, clear and precise information for the clinical diagnosis.
Surface chemistry and sample treatment
Surface biofunctionalization is crucial in biosensor development. To achieve high detection sensitivity, selectivity and reproducibility, the target bioreceptor must be appropriately anchored to the sensor transducer. Parameters such as the receptors’ density and orientation and the anti-fouling properties of the sensing surface must be carefully controlled to enhance the biosensor’s analytical performance and ensure its operational reliability.
 Examples of nanophotonic biosensors integrated in microfluidic systems. [Top: Courtesy of the authors / Bottom: Courtesy of Microfluidic ChipShop GmBH]
Examples of nanophotonic biosensors integrated in microfluidic systems. [Top: Courtesy of the authors / Bottom: Courtesy of Microfluidic ChipShop GmBH]
Numerous surface-chemistry strategies—such as covalent binding to functional monolayers of alkanethiols or silanes, or employing affinity-ligands such as the biotin–streptavidin pair—are widely used to accomplish these goals. However, these procedures need to be optimized for each specific application, depending on the nature, size and properties of the bioreceptor and the target analyte. Some new research perspectives envision overcoming those pitfalls through a universal-surface chemistry strategy, suitable for the detection of different types of biomarkers, like proteins, nucleic acids and pathogens. The development of multifunctional macromolecules, synthetic polymers and 2D materials could offer interesting alternatives to serve as the interface for bioreceptor immobilization.
Finally, the “nightmare scenario” of evanescent-field biosensors working in label-free format—and, yet, a key ultimate goal of such sensors—is direct analysis of human samples, like blood or urine. The large concentration of non-relevant molecules present in these fluids, together with the huge variability of samples from different patients, generally results in false-positive or false-negative analytical results. The solution to this challenge may lie in combining efficient anti-fouling surface coatings with integrated on-chip sample processing (for example, filtration membranes) that could be automated to ensure a rapid, reliable analysis.
Toward accurate, informative and early diagnosis
The vast majority of the clinical diagnosis applications developed for optical biosensors target the quantification of disease-specific circulating biomarkers. Classic examples are the detection of C-reactive protein (CRP) and cytokines, like interleukins or interferons, for inflammatory disorders, or of the carcinoembryonic antigen (CEA), the prostate specific antigen (PSA) and the p53 protein (among others) for cancer diagnosis. Also, DNA mutations of specific genes, like BRCA1 and BRCA2, have been extensively targeted for the detection of breast cancer. Still other optical biosensors, developed for infectious diseases, often target direct detection and identification of the pathogen, such as the bacteria Escherichia coli or Staphylococcus aureus.
 M. Cruz Cardenosa-Rubio (left) and Olalla Calvo-Lozano (right) working with a small benchtop SPR system developed in the laboratory. [Maria Soler]
M. Cruz Cardenosa-Rubio (left) and Olalla Calvo-Lozano (right) working with a small benchtop SPR system developed in the laboratory. [Maria Soler]
Recently developed photonic biosensors aim at still more advanced and improved diagnosis, targeting novel biomarkers for accurate, early disease detection and providing specific information related to prognosis and optimum therapy. We close this feature by looking at a number of areas where nanophotonic biosensors could help change the game.
MicroRNAs. MicroRNAs are small, non-coding nucleic acids that circulate in body fluids in very low concentration, but whose specific levels are strongly associated with pathological disorders. Direct detection and quantification of microRNA panels with multiplexed optical biosensors could become a highly precise screening technique for the early diagnosis of several cancers.
Some first steps have already been made toward that goal. A bimodal waveguide (BiMW) interferometric sensor has achieved ultrasensitive detection of such small nucleic-acid chains, identifying microRNA-181 in urine of bladder cancer patients, with a limit of detection of only 23 aM in less than 20 minutes in a direct assay. Micro-ring resonator arrays have also been employed for the multiplexed analysis of eight microRNAs from surgical glioma cells.
Autoantibodies. Likewise, new protein biomarkers have been described for early cancer diagnosis. Solid tumors in particular, like lung, breast or colorectal cancer, can trigger immune reactions in the patient, releasing specific antibodies into the bloodstream. These so-called autoantibodies appear and increase their levels at the initial stages of tumor development, even before the physiological onset of the illness. Nanoplasmonic biosensors have recently been developed to target different cancer-related autoantibodies, achieving adequate limits of detection in serum and plasma samples.
Bacterial detection. Infectious-disease diagnosis could present still another opportunity for optical label-free biosensors. Bacterial infections, if not promptly identified and suitably treated, can be extremely dangerous, in some cases even ending with a sepsis shock, in which the infection rapidly spreads to the whole body, a fatal development for the patient. Sepsis is actually the main cause of death in intensive care units (ICUs), with a mortality of 40%–50% due to delayed diagnosis and inappropriate antibiotic therapy.
Nanophotonic biosensors are fast becoming a versatile, powerful technology with exceptional potential for upgrading diagnostics—and improving global health care and well-being.
Biosensors able to detect and precisely identify a bacterial infection in a few minutes are essential to minimize this burden. Further, the ideal biosensor should be able to analyze in situ the specific bacteria, and provide information on possible antibiotic resistance, giving clues for the correct treatment. Here, ongoing research has already achieved significant results. Nanophotonic biosensors have been reported for bacterial analysis (for example, for S. aureus) with limits of detection between 4 and 10 colony-forming units (cfu) per milliliter. These sensors can even accurately differentiate methicillin-resistant (MRSA), methicillin-susceptible (MSSA) and borderline oxacillin-resistant (BORSA) S. aureus strains in less than 20 minutes. Other reported nanoplasmonic sensors can work in multiplex format, identifying two or more different bacteria species.
Routine monitoring. Finally, portable, user-friendly point-of-care biosensors might be very interesting for routine therapy monitoring and follow-up. Nanoplasmonic biosensors have been developed for monitoring of the drug acenocoumarol, an anticoagulant employed for thromboembolic disorders, and for adalimumab, prescribed for Crohn’s disease, as well as for follow-up on gluten-free diets for patients with celiac disease.
As these examples suggest, nanophotonic label-free biosensors are fast becoming a versatile, powerful technology with exceptional potential for upgrading diagnostics—and thereby improving global health care and well-being.
Maria Soler, Olalla Calvo-Lozano, M.-Carmen Estevez, and OSA Fellow Laura M. Lechuga are with the Nanobiosensors and Bioanalytical Applications Group, Catalan Institute of Nanoscience and Nanotechnology, Barcelona, Spain.
References and Resources
-
C. Rivet et al. Chem. Eng. Sci. 66, 1490 (2011).
-
G.A. Lopez et al. Nanophotonics 6, 123 (2017).
-
J.F. Masson. ACS Sens 2, 16 (2017).
-
M. Angelopoulou et al. Chemosensors 6, 52 (2018).
-
M.A.G. Porcel et al. Opt. Laser Technol. 112, 299 (2019).
-
E. Mauriz et al. Analyst 144, 7105 (2019).
-
C.S. Huertas et al. Front. Chem. 7, 724 (2019).
-
M. Soler et al. Expert Rev. Mol. Diagn., 1, 71 (2019).
-
C. Chen and J. Wang. Analyst, doi: 10.1039/C9AN01998G (2020).

