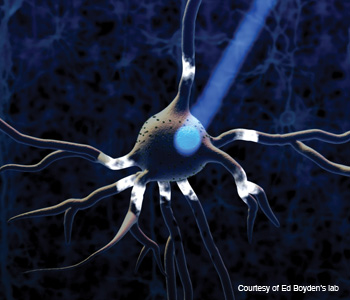
 A neuron with nanometer-sized proteins (tiny dots) on it flashes when blue light hits it.
A neuron with nanometer-sized proteins (tiny dots) on it flashes when blue light hits it.
The brain is the most mysterious and complex biological structure known. Human brains have in the neighborhood of 100 billion cells, and, at any given moment, multitudes of those cells are firing in intricate patterns and networks: Some excite other neurons, while others inhibit them.
Because of the complicated structure and functioning of the brain, much of what goes on within it remains unchartered territory. Despite much promising research, neuroscientists have only scratched the surface in their understanding of brain disorders such as Parkinson’s disease, epilepsy, blindness, anxiety and depression.
But thanks to the emerging field of optogenetics, that may be starting to change. By applying tools from optics, virology and genetics, researchers are literally shining light into the brains of animal subjects to observe and control how neural circuits work. Their work may inspire targeted new treatment options for many neurological conditions.
According to Karl Deisseroth, a professor of bioengineering and psychiatry and behavioral science at Stanford University and one of the pioneers of optogenetics, the field combines optics with genetics to control the functioning of well-defined events within biological tissues.
Deisseroth and Ed Boyden copioneered the field in a side-by-side collaboration while both were working on Ph.D.s at Stanford. Boyden is now a professor at the Massachusetts Institute of Technology’s Media Lab and a member of the McGovern Institute for Brain Research.
|
Their initial goal was to find a way to turn on or off specific cells within the brain. This required finding a way to make cells sensitive to being controlled, and then figuring out how to deliver the energy to achieve this.
“Neurons are electrical devices, so the first thing we thought about was what kinds of molecules we could put into a cell to make its electricity controllable by some form of energy,” says Boyden. The team already knew that many cells in nature contain molecules that respond to light—such as the plant cells responsible for sensing light and converting it into stored energy through the process of photosynthesis. When activated by light, these molecules allow charged ions to flow into the cells, setting up an electrical potential difference.
“The core tools of optogenetics—genes that encode light-activated regulators of ion flow from microbes—had been well known to exist for decades,” Deisseroth says. However, “nobody thought it would work to introduce them into neurons as a control tool.”
Evolution of optogenetics
Deisseroth and Boyden began their quest in 2000 by surveying the natural world for molecules that would work well in neurons. After four years of searching, they discovered a light-sensitive protein from algae called an “opsin” that had the right speed and biocompatibility to function in brain cells (neurons), which are famously finicky to work with. Algal cells use these opsins, which sit in their membranes, to sense light so they can navigate toward the surface of a body of water. The opsin molecule essentially acts as a tiny solar cell.
The next step for the team was to figure out how to insert opsins into neurons. “Thankfully, this molecule is completely encoded for by a snippet of DNA,” says Boyden. The team developed a procedure to insert the DNA—i.e., the gene for the opsin—into a virus and then inject the virus into neurons so that the brain cells could code for (produce) the opsin protein themselves and become light sensitive.
The researchers have several tools for delivering genes to the specific neurons they want to target. They can tune the virus used to carry the DNA so that it will only infect one type of neuron, for example, or they can insert genetic “regulatory” elements into the virus that will allow the protein to be expressed only in the target cells (even though it infects all the cells).
This first opsin to be used in optogenetics is known as channelrhodopsin-2 (ChR2), and it is turned “on” (excited) by blue light. In 2005, Deisseroth’s Stanford team used ChR2 to activate neural cells in vitro. They refined their technique, which is now widely used in labs all over the world, to enable them to deliver light directly to the brains of mice via a fiber-optic cable. The light selectively turns on specific groups of neurons, allowing reliable, millisecond-precision control over the cells’ patterns of firing action potentials—the voltage impulses that enable one neuron to convey information to another.
There’s an amazing demonstration of blue light excitation in action from the Deisseroth lab widely available on the Internet (www.youtube.com/watch?v=88TVQZUfYGw). In it, a lab mouse is shown with a fiber-optic cable inserted into the motor control center of its brain’s cortex, which controls its left-side movement; a thin fiber-optic cable runs out of its skull. When the light is turned on, the cable shines blue light, and the mouse circles left. When it is switched off, the mouse stops and returns to its normal behavior.
In other rodent studies, the Stanford and MIT researchers have used other channels that enable red light to excite specific groups of neurons, as well as pumps that allow yellow and red light to be used to inhibit them. Red light is better tolerated by tissues than light of shorter wavelengths because it scatters less and penetrates more deeply.
The researchers have now done animal studies covering the entire visible spectrum. “This enables us to do combinatorial work,” Deisseroth says. In other words, by studying various types of cells using different kinds of light, scientists can investigate more than one neural circuit to glean information about how various types of cells and pathways interact with one another.
 Boyden in the lab.
Boyden in the lab.
Having the ability to target specific neurons also enables systems neuroscientists to test causal rather than correlative hypotheses about the functioning of brain cells, explains Deisseroth. For example, it’s been long known that dopamine neurons fire when animals receive food or other rewarding stimuli. However, investigators did not know whether that was because those neurons directly triggered the experience of reward or because they were just passively reporting it. By exciting the dopamine neurons with light, the researchers showed that these neurons can in fact cause the reward sensation.
“Instead of looking for correlations or simply observing systems, we can now cause or inhibit specific events to control resulting phenomena,” Deisseroth says.
In 2007, Deisseroth and Boyden—who at that time were working separately—both independently showed that another light-sensitive protein, halorhodopsin, could inhibit the activity of neurons when it is illuminated with yellow light. However, it took several years and many molecular modifications to demonstrate that this pump could be successfully used in mice to optogenetically inhibit their behavior, as reported by the Deisseroth lab in Science in December of 2010.
Meanwhile, Boyden’s group continued its search for other opsins. They discovered multicolor silencers (inhibitors) that were an improvement over the old tools because they were more powerful and allowed for independent control of multiple kinds of neurons. The multicolor silencers are blue and red—which means that two different neurons can be differentially inhibited by blue light if they bear the two molecules. Boyden’s group described 100 percent neural silencing in mice that were awake in Nature in January 2010.
Boyden dubbed these new opsins “super silencers,” because they’re capable of completely silencing different sets of neurons with various colors of light. Simply put, the electrical activity of the neuron can be reduced to zero through light activation. By “shutting off” certain cells, researchers can better understand how neurons communicate with one another, which ones contribute to disease states, and how neurological signals orchestrate biological or behavioral functions.
Boyden’s super silencers were developed from two genes found in various natural organisms, including bacteria and fungi. These genes, called Arch and Mac, encode for light-activated proteins. Light activates the proteins, which then lower the voltage in the neurons and safely and effectively prevent them from firing. Neurons that are engineered to express halorhodopsin are specifically silenced by red light, while those expressing Mac are silenced by blue light.
Arch-expressing neurons can be switched off with greater precision and recovered faster than halorhodopsin-expressing neurons, notes Boyden.
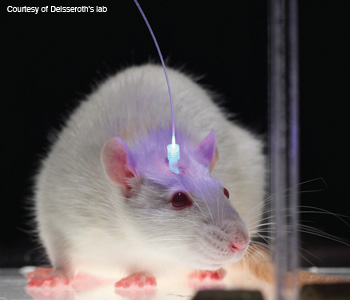 Mouse movement control demonstration: The lab mouse turns to the left when light activates the genetically engineered cells in its brain's cortex that control left side movement.
Mouse movement control demonstration: The lab mouse turns to the left when light activates the genetically engineered cells in its brain's cortex that control left side movement.
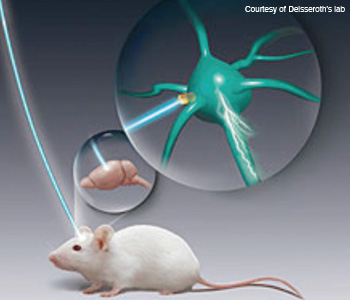 Making neurons react to light: Neuroscientists insert opsin genes into brain cells with the help of engineered viruses. They can then trigger these cells on demand with pulses of light and observe the effects on experimental animals' behavior.
Making neurons react to light: Neuroscientists insert opsin genes into brain cells with the help of engineered viruses. They can then trigger these cells on demand with pulses of light and observe the effects on experimental animals' behavior.
The next phase
Many research groups are now driving optogenetic technology forward—and part of this process involves moving beyond mice, whose brains contain only about 100 million cells. Recent studies have moved to larger-brained animals such as rats and monkeys. “We’ve done some work in monkeys now,” says Deisseroth. “It was a lot of work in terms of controlling cells very well,” he says.
As the field evolves toward studying more complex brains, “there might be a need for additional strategies to control more cells or to control a greater fraction of cells,” he continues.
One current area of focus at Deisseroth’s lab is building hardware for light delivery to living tissue that improves upon the fiber-optic system his team developed using laser diodes in 2007. They are now looking to develop fiber bundle strategies and digital micromirror methods for delivering complex light patterns into the brain.
Meanwhile, Boyden’s team is working to improve the technology on several fronts. For starters, they are continuing to look for new opsins by assessing molecules from organisms from all over the world—relying heavily on imaging in their search. For example, they use ion-sensitive dyes to help characterize opsins to figure out which ones will result in the right cellular change.
“We’re improving the molecules by multiple-fold every six months or so,” says Boyden. “Things are changing very rapidly now.” The team is working on finding new molecules that are even more color-shifted (i.e., red-light sensitive) and that are produced more quickly on the cell membranes when inserted into neurons. More red-light-sensitive molecules are desirable because red light travels further into the brain than blue light.
Boyden’s team is also developing new delivery mechanisms. Last year, he and his colleagues built probes that could be inserted deep into the brain, emitting light from many points all along the probe. The new probe includes many parallel optical fibers running in a band together, and each of the tiny waveguides terminates in a different depth along the length of the probe.
“An optical fiber might only emit light out of the very end, and therefore can only deliver light to one point in the brain,” explains Boyden. “But these probes can be inserted into the brain to deliver light to dozens and dozens of points all along the insertion axis of the probe.”
This technology is useful for many reasons. First, it could eventually be used to control neural circuits in three dimensions in the brain. It could also have clinical advantages over existing nonspecific methods; for example, it could be used for deep brain stimulation if the field of optogenetics evolves to the level of medical treatment in humans.
About two years ago, Boyden and colleagues published a study on the use of optogenetics in primate brains. They demonstrated that the light-sensitive molecules were being safely produced and that they could activate neural responses over periods of many months—in other words, they were fully functional. That wasn’t necessarily obvious or expected: Because these molecules were taken from algae, bacteria and fungi, it was possible that the primates could have reacted negatively to them.
The researchers have not yet been able to demonstrate optically driven effects on behavior in primates. However, optogenetics is still a new field and there is clearly more work to be done.
Right now, Boyden is focusing on developing a “super light delivery” probe that would serve as a 3-D light delivery device. “I think this will allow us to really start to understand how neural circuits work together across the entire brain to implement computations,” he says. Light can illuminate about a cubic millimeter at the end of the optical fiber that Boyden is developing. The light powers required are 0.10 mW/mm2. His team is currently working to invent multisite illuminators.
 Karl Deisseroth
Karl Deisseroth
Implications for neuroscience
The use of genetically engineered opsin proteins in the mammalian brain is revolutionizing neuroscience, according to Anatol Kreitzer, an investigator at the Gladstone Institute of Neurological Disease, and assistant professor at the University of California, San Francisco. Kreitzer has collaborated with Deisseroth on Parkinson’s disease research.
He is using optogenetics to define the function of certain types of cells in the brains of mice and to also identify specific neurons during electrical recordings from the brain. These recordings are obtained by placing a wire inside the brain, and recording the electrical signals from neurons located at the end of the wire. The wire transmits electrical signals to an amplifier, digitizer and computer. “We can identify cells during recordings by measuring whether they are light responsive,” Kreitzer says.
Using optogenetics properly requires a lot of equipment and expertise, says Kreitzer, although he believes it’s relatively straightforward for researchers who are already using the techniques of molecular biology, electrical recording or sophisticated behavioral paradigms.
According to Kreitzer, the most significant benefit of optogenetics for neuroscientists lies in its ability to noninvasively control chemical or electrical signaling in cells. “In the brain, this means gaining the ability to perturb specific kinds of cells, both to identify them during recordings and to test their function—for example, to test whether certain kinds of cells are necessary or sufficient for a given behavior,” he says.
Kreitzer experimented with mice that lack dopamine, which is a brain chemical that is greatly reduced in people who suffer from Parkinson’s disease. When he used optogenetics to activate the neural pathways in mice that regulated their movement, the mice were able to move around normally—even in the absence of dopamine. In fact, he was able to restore all of the motor deficits using this treatment.
His work illustrates how the selective stimulation of motor planning circuitry can play an important role in treating Parkinson’s and other disorders that involve the same neural circuits, including Huntington’s disease, Tourette’s syndrome, obsessive-compulsive disorder and addiction.
Use in humans?
Although many people assume that optogenetics is mainly being explored as a potential therapeutic device for humans, that is not Deisseroth’s main goal. “What’s most exciting about this research is that it can be used as a tool to probe and understand how neural circuits work,” he says. “If there were some clinical significance, that would be nice, but it will be dwarfed by the basic science significance.”
Indeed, optogenetics may turn out to be more useful as a model for how brain disorders arise—which could inspire other effective treatments—than as a therapy in and of itself. “It’s as if the percolation of activity is finely balanced and when the system is tipped too far one way or the other, activity spreads too far or not far enough—and disease symptoms result,” says Deisseroth.
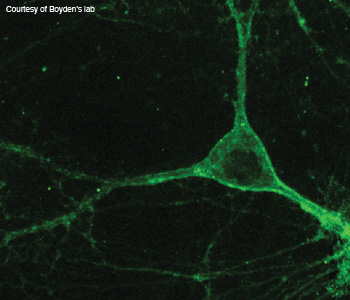 "Super silencers": This image shows a neuron expressing a genomically mined silencer.
"Super silencers": This image shows a neuron expressing a genomically mined silencer.
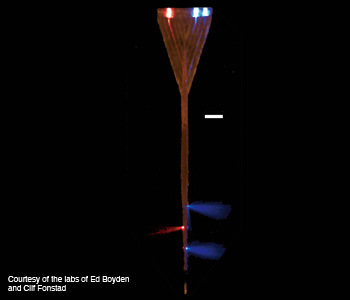 Brain probe light delivery system: A multiwaveguide brain probe delivers light to dozens of points along the axis of the device.
Brain probe light delivery system: A multiwaveguide brain probe delivers light to dozens of points along the axis of the device.
There are, however, other scientists who believe that optogenetics should ultimately be applied to human brains if it is found to be safe and effective, Boyden points out. “People are definitely trying them out, assessing the efficacy and safety in different systems in the brain, and using this toolset to discover principles of how to tackle brain disorders,” he says.
Boyden says that a couple of groups are pushing pretty hard right now to develop direct human therapies using optogenetics—one in Switzerland and one in Los Angeles, Calif., U.S.A., both of which are researching blindness. In addition, there’s another group at Case Western Reserve University in Ohio, U.S.A., that just spun out a company aimed at using optogenetic tools to address spinal cord injuries.
Boyden serves as an advisor to the LA team, and he distributes reagents to the other teams. “These three groups have publicly declared their intent to walk down that translational path,” he says. “Whether it’s a couple of years or many years depends on how the science goes, of course.”
The road ahead
For Deisseroth, the most rewarding aspect of his work in optogenetics has been witnessing how much it has helped researchers and how excited they are when they come for a training session with him. He is also gratified by how quickly optogenetic tools have been translated into versatile applications across many fields of biology. “In some ways, almost all I had hoped for optogenetics has already been achieved,” he says. “It’s become a standard part of biologists’ toolkit, with thousands of researchers benefitting, and it’s helping us to understand neurological and psychiatric disease more deeply.”
Deisseroth appreciates the relative straightforwardness of the optogenetic approach. He says that the core techniques are surprisingly easy to use once scientists are taught how to perform them. Light gives optogenetics a component of simplicity—there is no need to add chemicals, multiple genes or other complications to stimulate a response.
It’s not outrageously expensive either. According to Boyden, a complete optogenetics setup costs as little as a few thousand dollars for all of the required hardware and consumables.
Boyden has enjoyed learning from the people who are using these tools in their research. “Some people walk in and say, I’ve got this amazing problem where we’d like to turn off this part of the brain so we can turn off chronic pain or shut off a seizure,” he says. “I’m learning about all these problems that need solving, and you can be sure I’ll use this knowledge to decide what to invent next…”
Boyden predicts that optogenetics will become a routine biological research tool within the next 10 years. “The scope will also expand, and people will be developing methods for controlling all sorts of functions with light,” he predicts.
Deisseroth notes that optogenetic tools are already becoming standard in the study of neuroscience, operating in more than 1,000 laboratories worldwide.
Both Deisseroth and Boyden underscore the importance of supporting public and private funding for undirected basic science research. No one could have imagined at the outset where optogenetics research would go—or how it may continue to evolve. With the potential to illuminate how neural circuits work and inspire treatments for Parkinson’s disease, blindness, depression and many other disorders, optogenetics may help scientists to unveil the secrets of one of nature’s biggest mysteries: the human brain.
Sally Cole Johnson is a freelance journalist based in New Hampshire who specializes in optics, photonics, semiconductors and physics.
References and Resourcess
>> Ed Boyden's TED talk about the field of optogenetics: www.ted.com/talks/ed_boyden.html.
>> MIT Medial Lab: www.media.mit.edu.
>> Kart Deisseroth's Lab: www.stanford.edu/group/dlab.
>> Faculty of 1000 Post-Publication Peer Review report on the history of optogenetics: http://f1000.com/reports/b/3/11.
>> E.S. Boyden et al. "Millisecond-timescale, genetically targeted optical control of neural activity," Nature Neurosci. 8, 1263-8 (2005).
>> V. Gradinaru et al. "Optical Deconstruction of Parkinsonian Neural Circuitry," Science 324, 354-9 (2009).
>> X. Han et al. Neuron "Millisecond-timescale optical control of neural dynamics in the nonhuman primate brain," 62(2), 191-8, 30 (2009).
>> L. Buchen. "Neuroscience: Illuminating the Brain," Nature 465, 26-28 (2010).
>> Editorial. "Method of the Year," Nature Methods 8(1): 1-1(2010).
>> A. Kreitzer et al. "Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry," Nature 466, 622-6 (2010).
>> I.B. Witten et al. "Cholinergic interneurons control local circuit activity and cocaine conditioning," Science 330, 1677-81(2010).
