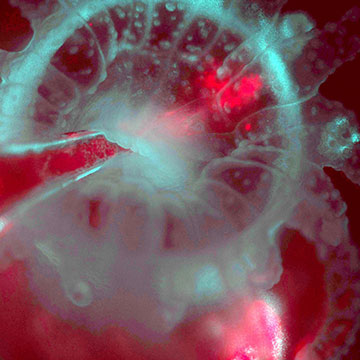
Using the L-SPI microscope to visualize the re-infection of an Astrangia polyp (cyan from reflected light) with algae (red from chlorophyll fluorescence), injected by pipette into the mouth of the polyp. [Image: Loretta Roberson]
In the laboratory, some photosensitive corals exhibit their own version of the observer effect: Exposing them to enough light for researchers to view them at microscopic scales changes the rates at which they grow and interact with symbiotic organisms such as algae. Scientists, however, want the coral polyps in their aquariums to act just as they would in the oceans.
To avoid disturbing their sensitive subjects, scientists in the United Kingdom and the United States have developed a light-sheet illumination system for performing fluorescence microscopy on coral polyps in captivity (Sci. Rep., doi: 10.1038/s41598-020-67144-w). Restricting the illumination to the focal plane of the microscope setup allows researchers to study the live creatures for extended time periods with minimal exposure to light.
Why corals matter
Corals are invertebrate animals that don’t move around, but rely on ocean currents and tiny symbiotic creatures to bring them food. Many, but not all, corals form reefs that host such diverse marine lifeforms that scientists call the reefs “rainforests of the ocean.” Reefs, however, are increasingly threatened by pollution, ocean acidification, increasing water temperature, photobleaching, overfishing and other factors.
Scientists can learn only so much from taking pictures of the exoskeletons that corals have left behind, or from fixing slices of tissue on a microscope slide. By bringing living corals into the laboratory and studying their skeleton-forming processes and their interactions with the single-celled organisms that cooperate with them, researchers hope to gain a better understanding of the life cycles and bodily functions of these important members of the ecosystem.According to Philippe Laissue, a bioimaging specialist at the University of Essex, U.K., traditional microscopes focused on live corals either provided pictures of “underwhelming quality” or harmed the corals under study. When the university’s coral reef researchers sought his advice on laboratory studies, Laissue realized that light-sheet fluorescence microscopy—also known as selective plane illumination microscopy, or SPIM— would provide the happy medium the biologists were seeking.
Widening the light sheet

A coral polyp (Astrangia poculata) in the L-SPI sample chamber with tentacles fully extended. [Image: Loretta Roberson]
Light-sheet microscopes illuminate their subjects by sending the light to the subjects from the side, perpendicular to the direction of observation. Often the light comes from a laser whose beam is first expanded and then focused through a cylindrical lens. At any one moment, the narrow light sheet stimulates fluorescence in only a thin slice of the sample.
Unfortunately, Laissue found that commercially available light-sheet microscopes could not accommodate samples as big as a coral polyp, so he would need to develop his own SPIM system. “Before this project, I had used many microscopes, but never built one myself,” he says. “So getting into the technicalities and optics was at first a little daunting.”
Guided by tutorials on the OpenSPIM website—a DIY guide to the technology published by researchers at the Max Planck Institute of Molecular Cell Biology and Genetics in Germany—Laissue built an experimental device called a large selective plane illuminator, or L-SPI. Its light path started with a 488-nm-wavelength semiconductor laser. After the beam was expanded twice, a rotating mirror—taken from an old laser printer—fanned the beam out into a sheet. The light sheet was 20 mm wide and had a “waist” (the thinnest measurement in the direction of propagation) of varying sizes, from 20.7 μm to 11.7 μm. The wider waists enabled views of a whole colony of polyps, whereas the thinner waists allowed the researchers to focus on a single polyp and its surrounding autofluorescent tissue.“Compared to many other custom-made microscopes out there (and featured in OPN), it is a very simple setup,” Laissue says of the OpenSPIM plans. “To me, simple means robust, easy to use and easy to replicate.”
Applying the light sheet
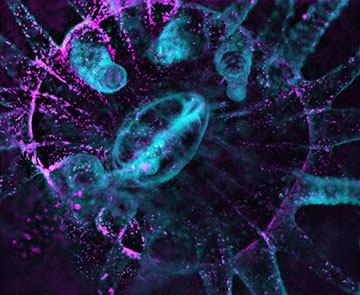
Dark-field imaging is used to reveal the non-autofluorescent coral tissue in Astrangia poculata, a species with no intrinsic fluorescence. Non-fluorescent tissue (cyan) and dinoflagellates (magenta) are combined in the image. [Image: Philippe Laissue, Scientific Reports (2020)]
Laissue and his colleagues first turned their L-SPI onto samples of Acropora muricata, or staghorn coral, which forms small, fast-growing colonies and are the most photosensitive coral species known to science. The team showed that the instrument could perform fluorescence imaging of the corals for several hours at a time, and they quantified the irradiance levels at which blue light started to damage the coral tissue.
Since Laissue is also affiliated with the Marine Biological Laboratory (MBL), USA, he collaborated with MBL biologist Loretta Roberson on studies of Astrangia poculata, a coral species found widely distributed in the Atlantic Ocean and Caribbean Sea.
Laissue says he hopes the new imaging technology can shed more light on the basic biology of corals and the triggers that cause the mutually beneficial relationships between corals and microorganisms to break down. Biologists have recently discovered that an illness called “stony coral tissue loss disease” is ravaging corals in the Caribbean, and he says that “gentle live imaging over long periods” may help scientists treat and prevent the disease.
“In a wider sense,” he adds, “this project has really opened my eyes to phototoxicity and the often damaging effect that fluorescence excitation light can have on living samples, so we are raising awareness of this (potentially very misleading) phenomenon and hoping to set some standards on assessing, reporting and minimizing phototoxicity in other live-imaging approaches.”
The authors also posted a video summary of the work online:
