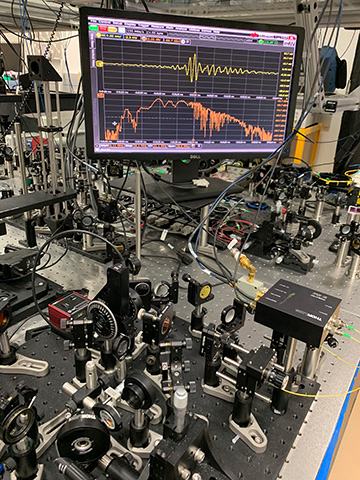
NIST's tabletop frequency comb apparatus. On the monitor, the yellow line is the electric field of mid-IR laser light that has passed through air in the lab, and the orange line shows the corresponding spectrum of the light, with dips corresponding to the colors absorbed by water detected in the air. [Image: NIST]
Frequency combs have found numerous applications in physics, from ultra-precise timekeeping to the search for extrasolar planets. Scientists at a U.S. laboratory have built a compact mid-infrared frequency comb apparatus that can rapidly identify molecules of biological and chemical interest (Sci. Advances, doi:10.1126/sciadv.aaw8794).
Mid-IR light, near-IR sampling
The infrared (IR) spectral region between 2 μm and 20 μm has long been recognized as the “molecular fingerprint” region because all organic and many inorganic molecules exhibit rotational and vibrational resonances at those wavelengths. Recent advances in mid-IR laser sources for spectroscopy have spurred research into new sensing techniques for detecting diseases and contaminants (see “Mid-IR Spectroscopic Sensing,” OPN, June 2019).
The team from the U.S. National Institute of Standards and Technology (NIST), led by OSA Fellow Scott Diddams and postdoctoral fellow Abijith Kowligy, employed two mutually phase-locked erbium fiber lasers to generate both mid-IR light and near-IR sampling pulses. This setup reduced sampling and jitter errors that had limited previous attempts to extend dual-comb sampling from terahertz to mid-IR frequencies.
Because of the near-IR sampling, the researchers could detect the signal with a room-temperature semiconductor photodetector, rather than a cryogenically cooled photodetector. Thus, the team says, the apparatus takes up less than half a square meter of benchtop space. The experiment measured spectra between 3 μm and 27 μm (370 to 3,333 cm–1) with spectral resolution of up to 0.003 cm–1.
Tackling complex compounds
After measuring bending vibration lines from simple molecules such as carbon dioxide and ammonia, the NIST group tackled two more complex compounds: a carvone (a terpenoid found in scented products) and a monoclonal antibody (specifically, one developed at NIST as a reference standard for the pharmaceutical industry). The researchers measured the spectral positions of the antibody's amide bands—key bonds that determine the folding and unfolding of proteins.
Researchers from the University of Campinas, Brazil, and the Institute of Photonic Sciences at the Barcelona Institute of Science and Technology, Spain, also contributed to the study.
