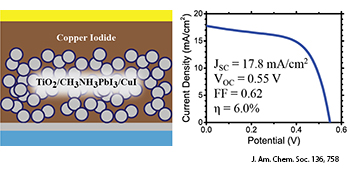
In this perovskite solar cell, gold tops an overlayer of a copper iodide, perovskite, a blocking layer (grey) and a fluorine-dope tin oxide substrate (blue).
Several recent studies have rapidly pushed perovskites to the fore as a promising material for inexpensive, high-efficiency photovoltaic cells. Perovskites, a class of superconductive materials with the same crystal structure as calcium titanium oxide (CaTiO3), exhibit a wide array of interesting properties. In particular, hybrid versions of lead halide perovskite (CH3NH3PbI3) and methylammonium lead halide perovskites are of interest for thin-film solar cells. They feature such properties as high charge carrier mobility, panchromatic absorption and long charge carrier lifetime, which enable photo-stimulated electrons and holes to travel further through solar cells without losing their energy.
The highest efficiencies of perovskite solar cells have exceeded 15 percent, but one challenge is the expense and low hole mobility of the organic materials used for hole conduction. A recent paper by Emilio Juarez-Perez and colleagues at the Universitat Jaume I (Castello, Spain) examined the role of the electron- and hole-selective contacts in the performance of hybrid lead halide perovskite (J. Phys. Chem. Lett., DOI: 10.1021/jz500059v). The team compared cells with different contact materials (TiO2, ZnO, and CdS) to cells without contacts to describe the specific role of each contact. They found that selective contacts are largely responsible for the high voltage in this kind of device, and that it mainly governs the recombination rate—an important implication to consider in future development of perovskite photovoltaics.
In another recent paper, Jeffrey Christians and colleagues at the University of Notre Dame (Ind., U.S.A.) identified copper iodide (CuI) as a possible inexpensive, inorganic alternative for a hole conduction material, achieving a stable power conversion efficiency of 6.0 percent (J. Am. Chem. Soc., DOI: 10.1021/ja411014k). Although the conversion efficiency of CuI still doesn’t beat the 7.9 percent of the best hole-conducting polymer, spiro-OMeTAD, the latter has a commercial price ten times that of gold and platinum. The researchers used impedance spectroscopy to reveal that CuI has an electrical conductivity two orders of magnitude higher than spiro-OMeTAD, so future improvements to the inexpensive CuI structures could enable solar-cell efficiencies of between 8 and 10 percent, which would make them a competitive value proposition.
