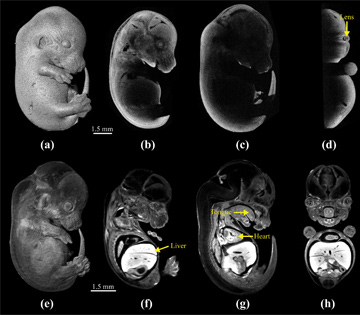
 Mouse embryo at 13.5 days post-coitum as imaged by OCT (a to d) and OPT (e to h). [M. Singh et al., Biomed. Opt. Express 7, 2295 (2016)]
Mouse embryo at 13.5 days post-coitum as imaged by OCT (a to d) and OPT (e to h). [M. Singh et al., Biomed. Opt. Express 7, 2295 (2016)]
Studies of disease etiology often rely on the murine (mouse) model, which provides valuable genotypic information. Yet associated phenotypic information from murine embryos is often lacking. Of the various techniques that have been proposed for imaging such embryos, optical modalities may provide the best compromise among speed, penetration depth and resolution. This year, we developed approaches to improve embryonic imaging with optical coherence tomography (OCT),1 and also provided a detailed comparison of OCT and another modality, optical projection tomography (OPT), in murine-embryo studies.2
Most commonly used for ophthalmological imaging,3 OCT has proved a powerful embryonic-imaging modality as well, as it can provide in vivo, 3-D imaging at resolutions of about 10 µm. Yet OCT’s short depth penetration limits its usefulness, especially in the later stages of embryonic development. We have overcome this limitation using multi-angle OCT imaging coupled with subsequent automatic image co-registration, which allows deep imaging of embryos.1 We have also used an ultrafast, phase-sensitive OCT system to demonstrate direct, video-rate 4-D imaging of embryo cardiodynamics—an improvement over previous methods that have relied on gating or synchronization, which are slow and error prone and require significant post-processing.4
A second modality, OPT, was developed nearly 15 years ago specifically for imaging embryos at high resolution.5 OPT overcomes traditional OCT’s imaging-depth limitation using multi-angle projection imaging and back-projection, and can also provide spatiotemporal functional analysis. Through its use of fluorescence, OPT provides structural and functional information with micrometer-scale spatial resolution and exquisite contrast, clarity and image quality. Yet, OPT also requires lengthy fixation and clearing procedures, which limit its use for in vivo imaging and which can even alter the embryo morphology—a fact we have quantified by directly comparing embryos imaged by OCT and OPT.2
We believe that our work in embryonic imaging will provide deeper insights into the correlation between genetic changes and phenotypic alterations, to help identify the disease origins, manifestations and efficacy of treatments.
Researchers
Manmohan Singh, Chen Wu, Raksha Raghunathan, Narendran Sudheedran and Kirill V. Larin, University of Houston, Texas, USA
Shang Wang, Victor Piazza, Tegy J. Vedakkan, Irina Larina and Mary E. Dickinson, Baylor College of Medicine, Houston, Texas, USA
References
1. C. Wu et al. J. Biomed. Opt. 21, 26002 (2016).
2. M. Singh et al. Biomed. Opt. Express 7, 2295 (2016).
3. D. Huang et al. Science 254, 1178 (1991).
4. S. Wang et al. Opt. Lett. 40, 4791 (2015).
5. J. Sharpe et al. Science 296, 541 (2002).
