![]()
A clinical trial of a treatment combining optogenetic modification and light-stimulating goggles restored the ability of a blind retinitis pigmentosa (RP) patient to distinguish objects and perform simple visual tasks. [Image: Getty Images]
Optogenetics—which involves the use of genetic engineering and light to control the function of nerve cells—has proved a revolutionary technique in the biology lab, allowing mapping of the neural networks of rodents and even primates. The technique has also tantalized researchers as a potential route toward therapies to treat certain neurophysiological diseases in humans.
Now, in early results from a clinical trial, a European research team reports that an experimental therapy combining optogenetics, gene therapy and specially designed, light-emitting goggles has restored partial visual function in a blind patient suffering from retinitis pigmentosa (RP). The study authors characterize the result as “the first reported case of partial functional recovery in a neurodegenerative disease after optogenetic therapy” (Nat. Medicine, doi: 10.1038/s41591-021-01351-4).
Devastating disease
RP is a rare genetic disease involving a breakdown and loss of the retina’s photoreceptor cells—the so-called rods and cones. In typical patients, it causes progressive erosion of eyesight beginning in the young adult years, with reductions in night vision and loss of some peripheral vision, and leads to blindness by around age 40.
Roughly 1 in 4,000 persons worldwide are thought to suffer from RP. And it’s a genetically complex scourge; mutations of any one of as many as 100 separate genes have been implicated in the disease. That has made it difficult to zero in on a direct cure.
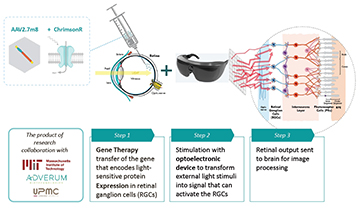
The group behind the current clinical trial—which includes scientists from several universities in France, the United Kingdom, Switzerland and the United States, as well as the Paris-based clinical-stage biopharma company GenSight Biologics—wanted to see whether optogenetic techniques could be tapped to lessen some of RP’s effects. Optogenetics involves the transfer of genes that encode light-sensitive proteins into neurons via gene therapy. This allows the neurons to be remotely fired, controlled or tracked using optoelectronically delivered light signals.
The GS030 system
The specific trial, dubbed the PIONEER Phase I/II study, is testing the GS030 optogenetics platform developed by GenSight and its academic partners. The system begins with a genetic modification of retinal ganglion cells, which are located near the inner surface of the retina. In a healthy eye, these cells serve as a sort of clearing house, transmitting visual signals sensed by the eye’s photoreceptors further on to processing centers in the brain.
The system under clinical trial involves genetic modification of retinal cells to encode light-sensitive proteins, coupled with optoelectronic goggles that directly stimulate the cells with light signals. [Image: Courtesy of GenSight Biologics] [Enlarge image]
The modification—delivered in a single injection via gene-therapy techniques—substitutes, into the retinal ganglion sequence, a gene that encodes for the light-sensitive protein ChrimsonR-tdT. That makes the ganglion cells directly responsive to bright, 590-nm (amber) light. The modification thus allows the ganglion cells to be stimulated directly by 590-nm photons, completely bypassing the photoreceptors in the eye that have been destroyed by RP.
The other component of the treatment is a medical device—specifically, a pair of optoelectronic goggles tuned to deliver light of the right wavelength based on visual stimuli.
The goggles capture images from the real world using a so-called neuromorphic, or event, camera. Unlike a conventional camera, these devices do not create a photograph-like image of a scene, but instead detect pixel-by-pixel changes in intensity. The goggles then map those events into monochrome images that can be projected onto the genetically modified retinal cells, in real time, via 590-nm pulses of light.
One patient’s results
While the PIONEER trial involves more than a dozen patients in multiple locations, the results reported in the new Nature Medicine paper come from only a single one of those test subjects. Before treatment, the 58-year-old male patient in question, who was first diagnosed with RP some 40 years ago, was able to distinguish only the relative presence or absence of light, and not to pick out shapes or objects.
The trial protocol isn’t a swift one. After the patient’s “worse-seeing eye” was injected with viral vectors containing the optogenetically modified genes, he was monitored for four and one-half months to confirm that there were no adverse effects, and to give the modified genes time to gain a foothold in the patient’s retina. After that, the patient participated in seven months of systematic visual training using the GS030 goggles.

In tests of the GS030 system, a blind RP patient, who before treatment could perceive only the presence or absence of light, was able to pick out, count and touch objects, such as the notebook shown here. [Image: Courtesy of GenSight Biologics]
After the training—and roughly a year after the initial gene-therapy treatment—the patient was ready for tests to analyze any visual improvement. First, the man was asked if he could perceive, locate and touch small objects (a notebook and a box of staples) that were set on a table by the researchers, and whether he could perceive, count and locate several small tumblers. In tests without the light-stimulating goggles, the patient could not make out any of the objects. In tests with the goggles, he was able to pick out, count and locate the objects between 36% and 92% of the time. Not surprisingly, the best results came when the subject was tasked with picking out the largest object (the notebook).
The researchers paired these tests with multichannel electroencephalography (EEG), which measures the level of neuronal activity across the brain. The EEG traces suggested, according to the team, that the retinal stimulation induced by the optogenetic therapy was propagating to the brain’s primary visual cortex, and shaping its activity.
On the street
Even more remarkable, perhaps, was the patient’s experience after leaving the indoor testing lab. The researchers write that outside, using the goggles, the man “spontaneously reported identifying crosswalks, and he could count the number of white stripes.” The patient also subsequently noted “major improvement in daily visual activities, such as detecting a plate, mug or phone, finding a piece of furniture in a room or detecting a door in a corridor, but only when using the goggles.”
The reported results are from only a single patient, of course, and don’t constitute a complete clinical trial. And there were some minor issues; for example, the patient apparently had a sense of “vertical vibrations” when perceiving an object, even though the object itself was not moving. Still, the preliminary result seems to constitute a striking potential vindication of optogenetics’ clinical potential for treating patients with RP—and, perhaps, other neurodegenerative diseases.
The research included scientists from the Sorbonne Université, the Institut de la Vision, the Centre Hospitalier National d’Ophtalmologie des Quinze-Vingts, the Fondation Ophtalmologique Rothschild, and GenSight Biologics, Paris, France; the University of Pittsburgh School of Medicine, USA; the Moorfields Eye Hospital and UCL Institute of Ophthalmology, London, U.K., and the Institute of Molecular and Clinical Ophthalmology Basel and University of Basel, Basel, Switzerland.