Researchers in China and Germany, inspired by the skin structure of the panther chameleon (shown here), have devised a method for creating programmably color-changing, biocompatible hydrogels for possible use in sensors and artificial skins. [Image: MarkBridger/Getty Images]
Taking a cue from the color-changing prowess of the panther chameleon, researchers in China and Germany have developed an artificial hydrogel that can generate programmable fluorescent color changes triggered by variations in temperature or pH (Cell Rep. Phys. Sci, doi: 10.1016/j.xcrp.2021.100417). The key to the system is a layered core–shell structure that surrounds a central, red-fluorescent hydrogel substance, or luminogen, with thin layers of green and blue hydrogel luminogens—the intensities of which can be controlled independently.
The team demonstrated the novel hydrogels in an application involving the detection of spoilage in seafood, in response to amine vapors given off by microbes as the fish goes bad. The group also envisions potential uses in anti-counterfeiting, dynamically camouflaged robots and other areas.
Dermal magic
As revealed in a study published in 2015, the panther chameleon achieves its dazzling color-changing feats through two skin layers, arranged in a core–shell structure, that are infused with guanine photonic nanocrystals. By independently tuning of the lattice spacing in these nanocrystal-bearing layers, the creature can achieve rapid, brilliant color changes—and thereby demonstrate its mojo in courtship or aggressive situations.
The researchers behind the new work—including scientists from the Chinese Academy of Sciences, the South China University of Technology and Tsinghua University, China, and the Karlsruhe Institute of Technology, Germany—wanted to see if they could cook up an artificial material that mimicked the chameleon’s dermal magic. They zeroed in on the prospects for achieving the feat by using a similar core–shell structure in polymeric hydrogels. These waterlogged, soft and biocompatible materials have the potential to integrate into a variety of applications.
Inside-out growth
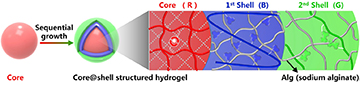
The team used an inside-out chemical process to grow tunable blue- and green-fluorescent layers on a red-fluorescent hydrogel core. [Image: W. Lu et al., Cell Rep. Phys. Sci., doi: 10.1016/j.xcrp.2021.100417 (2021); CC-BY-NC-ND 4.0] [Enlarge image]
To build their color-shifting core–shell hydrogels, the researchers looked to another natural example: the inside-out growth characteristic of eggs and onions. They began with a core containing a specially designed monomer, K6APA, that includes a chemical ligand that forms red-fluorescent complexes when linked up with ions of the element europium (Eu3+). They then bathed the core material in aqueous Eu3+ solutions, creating the desired fluorescent complexes and also packing the hydrous material with excess free europium ions.
Next, this red-fluorescent core hydrogel—referred to by the team as the “Eugel”—was incubated successively in solutions including sodium alginate (Alg) and blue- and green-fluorescent polymers. This created, around the Eugel core, an aqueous shell consisting of thin layers of fluorescent material around the Eugel. As the excess europium ions from the core diffused into this outer shell, they reacted with carboxylate groups in the sodium alginate, causing the Alg layers themselves instantly to gel up.
The result of the process was disk-like hydrogel blobs that included a red-fluorescent Eugel core and thin surrounding blue- and green-fluorescent Alg layers. The thickness of the blue- and green-fluorescent layers could be adjusted by adjusting the concentration of europium ions available in the red-fluorescent Eugel core. What’s more, because of the spectral overlap of the specific fluorophores, the team, by tweaking the Alg chemistry, could achieve continuous color changes when the material was exposed to UV light.
Sensing spoiled fish
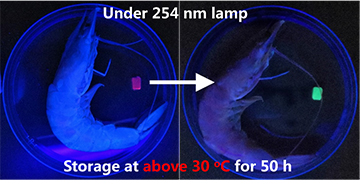
The researchers tested strips of their core–shell hydrogels as chemosensors to detect biogenic volatile amine vapors, a known indicator of fish spoilage. [Image: W. Lu et al., Cell Rep. Phys. Sci., doi: 10.1016/j.xcrp.2021.100417 (2021); CC-BY-NC-ND 4.0] [Enlarge image]
The team also found that, by mixing the sodium alginate of the Alg with different proportions of chemically responsive or thermoresponsive material, the Eugel–Alg structures could be pressed into service as temperature or chemical sensors. In one proof-of-concept demo, the researchers prepared a core–shell hydrogel with shell fluorescence that changed with increasing concentrations of amine vapors, such as ammonia. Such vapors are known to be an indicator of spoilage in fish and shrimp.
They then created test strips of the material and dropped them into packing boxes containing fresh fish and shrimp, which were then stored at different temperatures for two days. Sure enough, when the researchers returned, the hydrogel strip with fish stored at room temperature had changed color from a dull purple to a bright green—an indication of substantial amine vapor, and a clear sign that the seafood was no longer fit for dinner.
Toward dynamic skins for robots
In a press release accompanying the research, the team leader, Tao Chen, noted that the method for creating the core–shell multi-luminogen structures through ion diffusion “proves to be general.” That suggests, Chen says, that the method could expand for a wide range of applications in soft color-changing materials.
Such applications, the team writes, could include “chemosensing, bioimaging, information encryption, and biomimetic color-changing soft actuators and robotics.” Indeed, Chen notes that the team is currently working on “biomimetic soft camouflaging skins,” toward use in “desirable active camouflage, display and alarm functions in robots.”