
DESY researcher Wiebke Ewert points out a drug binding site (green) on an electron-density map of the SARS-CoV-2 main protease. A recent, high-throughput X-ray screening at the DESY PETRA III beamline uncovered several promising drug candidates against the protease. [Image: DESY, Christian Schmid]
In recent months, the talk in emails and Zoom calls has often turned to COVID-19 vaccines—who’s already gotten which of the jabs; who else might get one soon. But vaccines are only part of the anti-coronavirus equation. As with influenza, antiviral drugs for directly treating infections will prove crucial to living with COVID-19 in the long haul, and these drugs have been an active research quest in themselves.
Now, enabled by the giant PETRA III storage ring at the Deutsches Elektronen-Synchrotron (DESY), a group comprising more than 100 researchers in Germany and several other countries has zeroed in on one viral site that looks ripe for attack—and a number of potential drug compounds that could be up to the job (Science, doi: 10.1126/science.abf7945). Two of the drug candidates look sufficiently promising that they’re already under investigation in preclinical studies.
Targeting protein scissors
The work of the research team, led by Alke Meents of the Center for Free-Electron Laser Science at DESY, Hamburg, Germany, did not focus on new, as yet unknown treatment compounds. Instead, the team sought to cull through nearly 6,000 known active substances already approved for treatment of other diseases, to see if they might be repurposed somehow in the anti-COVID fight.

Lead researcher Alke Meents at the PETRA III beamline P11, in DESY’s Max-von-Laue Experimental Hall. [Image: DESY, Christian Schmid]
The researchers were looking in particular for compounds that might bind to the SARS-CoV-2 virus’ main protease (Mpro). The protease constitutes the protein “scissors” used by the virus to clip polyproteins translated from viral RNA in the host cell at specific sites, and thereby to enable new copies of the virus to be made. Finding a way to shut down Mpro could thus keep the virus from reproducing, and stop the infection in its tracks.
Determining whether a drug candidate might somehow be effective in blocking Mpro’s action requires looking in detail, via X-ray diffraction, at the structure of the drug proteins, to see if they might have the right “lock-and-key” geometry to fit a binding center on the protease. Such a geometry allows the drug to glom onto the Mpro surface, and potentially to prevent the protease from doing its work processing the viral proteins.
Finding the right molecular fit, in a population of close to 6,000 drug candidates, would mean doing structural biology on a grand scale. Indeed, a press release accompanying the work characterized the drug screening as “probably the largest of its kind.”
P11: Tuned for protein crystallography
To make this massive job tractable in a reasonable time, the researchers relied on the PETRA III light source at DESY. PETRA III is a 2.3-km-long synchrotron storage ring that is, according to DESY, the “most brilliant storage-ring-based X-ray radiation source in the world.” The team did much of the work at the facility’s P11 beamline, a station specifically tuned for structural biology and macromolecule crystallography. In an example of the specialized X-ray optics that have been developed for DESY facilities, P11 includes dynamically bendable mirrors and other optical elements that preserve beam coherence and deliver a high photon flux to a very small focal spot at the point of the sample.

The technical equipment at PETRA III’s P11 beamline includes a robotic arm that can execute fully automated sample changes. [Image: DESY]
These specialized X-ray optics allow the 3D structure of proteins to be identified with atomic precision. Also—in a feature that would prove crucial to the current work—P11 includes a robotic arm that can execute fully automated sample changes at a brisk pace. As a result, each of the more than 7,000 measurements made in the experiment reportedly took only around three minutes.
Efficient pipeline
The researchers—granted exclusive access to the DESY P11 beamline, as well as special access to the P13 and P14 beamlines at the facility, managed the European Molecular Biology Laboratory (EMBL), over a period of four weeks—put these specialized tools to work on thousands of active substances already used in the treatment of non-COVID diseases. The candidate substances were drawn from existing libraries at the Fraunhofer Institute for Translational Medicine and Pharmacology and at the Italian company Dompé Farmaceutici SpA.
A massive chemistry effort produced crystallized samples for the thousands of substances, which were then pushed through the PETRA III beamlines to generate raw X-ray diffraction images. Those images flowed directly into a custom-written software pipeline to automate processing and refinement of the raw diffraction data into structures that could be analyzed for a potential fit with the SARS-CoV-2 Mpro.
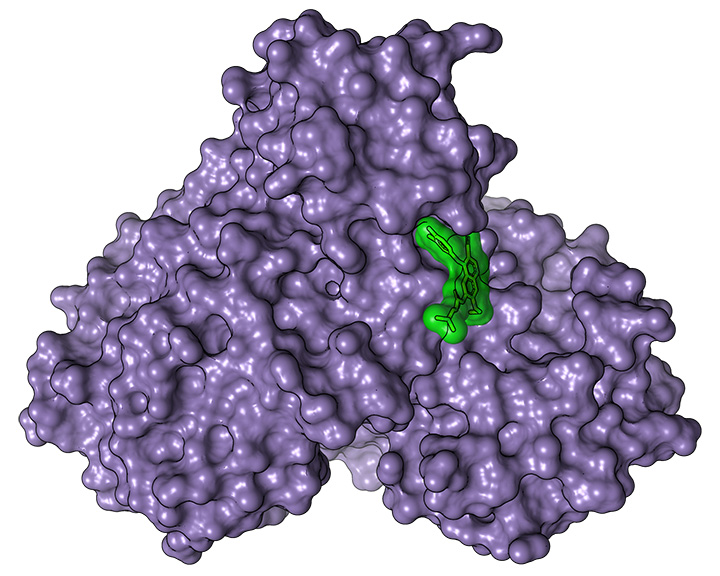
Surface representation of the structure of the main SARS-CoV-2 protease (violet) with the bound active substance pelitinib (green). [Image: DESY, Sebastian Günther]
This high-throughput workflow allowed the DESY team to narrow down the thousands of possible drug candidates to just 37 active substances that might potentially bind with the coronavirus protease. Next, research partners at the Bernhard Nocht Institute for Tropical Medicine, also in Hamburg, experimented with these substances in actual cell cultures, to see if they slowed down viral replication. Seven of them did—and two of them, Calpeptin and Pelitinib, showed a sufficiently compelling combination of antiviral action and cell compatibility that preclinical investigations for the drugs against COVID-19 are already underway.
Synchrotrons vindicated
In a press release accompanying the work, Helmut Dosch, the chairman of the DESY Directorate, maintained that the efforts at PETRA III “impressively show how relevant high-brilliance synchrotron light sources are for the development of future medicines and for health research as a whole.”
Indeed, in addition to the several potential anti-COVID treatment candidates uncovered in the work, the high-throughput study identified something else: an unexpected, and previously unknown, binding site on Mpro that could offer another drug target. Christian Betzel of the University of Hamburg, one of the initiators of the study, said that such a result “can really only be achieved at a synchrotron light source like PETRA III.”
