![]()
Researchers believe that work elucidating the structure of reflectin proteins in the skin of squid—such as Doryteuthis opalescens, shown here—could open possibilities for applications of these proteins in electronics, optics and medicine. [Image: Getty Images]
Cephalopods—a class of marine animals that includes squids, octopuses and cuttlefish—can readily change the appearance of their skin to blend into their surroundings or mimic other species. These stunning camouflage abilities come from optical components in the skin, such as light-reflecting cells and pigmented organs, along with an underlying musculature that alters the skin’s texture and shape.
The building block for many of these optical components is a unique protein called reflectin, which has proved troublesome to study due to its atypical chemical composition and sensitivity to the environment. Now, an international team of researchers has resolved the molecular-level structure of a reflectin variant by using experimental techniques that make the protein much easier to work with (PNAS, doi: 10.1073/pnas.2009044117).
“With this research, we have focused on developing a detailed fundamental understanding of how reflectins function at a molecular level,” said study author Alon Gorodetsky, associate professor of chemical and biomolecular engineering at the University of California, Irvine, USA, in a press release accompanying the research. The insights gleaned from the study, the team believes, could “afford new opportunities in biochemistry, cellular biology, bioengineering, and optics.”
A user-friendly reflectin variant
Previous studies that attempted to reveal the 3D structure of reflectin suffered from multiple drawbacks. Some required organic solvents or denaturants that damaged the natural structure, while others only investigated aggregated clumps of reflectin. Gorodetsky and his colleagues saw an opportunity to avoid these experimental pitfalls by choosing a more user-friendly reflectin variant based on a water-soluble parent protein found in multiple types of squid skin cells.
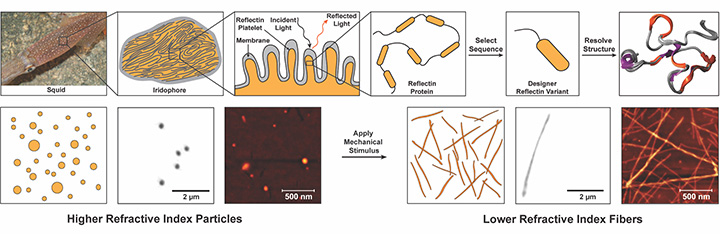
Through a combination of experimental and computational methods, researchers have revealed the 3D structure of a reflectin variant and developed methods for controlling its hierarchical assembly. [Image: Roger T. Hanlon (squid photo); Preeta Pratakshya / UCI (schematics)] [Enlarge image]
The team first made computational predictions of the variant’s structure by performing molecular dynamics simulations. Mapping of the conformational and energetic landscape verified that it could be probed successfully with experimental techniques, so the variant was subsequently produced and characterized with small-angle X-ray scattering.
To resolve the 3D structure of the protein, Gorodetsky and his colleagues employed nuclear magnetic resonance spectroscopy. They found specific pH values and protein concentrations that allowed the variant to remain in a monomeric state during measurement instead of aggregating together.
Linking structure with optical properties
Lastly, the researchers wanted to see if they could link the optical properties of certain architectures formed by the reflectin variant with their degree of structural order. After several hours of gentle mechanical agitation in water, unordered proteins exhibited signs of increasing ordered conformation into architectures like chains and sheets. Holotomography revealed that the degree of structural order correlated with the measured refractive indices.
“Through our synergistic computational and experimental approaches, we were able to elucidate the three-dimensional structure of the reflectin variant, thereby establishing a direct correlation between the protein’s structural characteristics and intrinsic optical properties,” said Gorodetsky. “This research can be viewed as a valuable conceptual framework for using this class of proteins in bioengineering applications.”
In one example of such an application, the researchers point out that reflectin variants, if expressed in mammalian cells, could enable the tweaking of “light-scattering properties and transparencies” in living organisms, making their cells and tissues “amenable to 3D visualization.” On the flip side, reflectin proteins could also, in the team’s view, find a place in optical materials, enabling the development of polymer–reflectin hybrids to create soft materials “3D refractive-index distributions that are not readily accessible via conventional fabrication techniques.” These, the authors suggest, could extend the use of polymers in some exotic optical applications that rely on gradient-index materials, such as flat lenses.