[Image: Getty Images]
As the COVID-19 pandemic spreads across the world, patients, technicians and scientists depend on state-of-the-art molecular-analysis instruments as they fight against SARS-CoV-2, the virus causing this disease. Optics and photonics technologies embedded in these instruments—such as high-quantum-efficiency multispectral cameras, visible-light laser diodes and LEDs, infrared bolometer arrays, narrowband optical filters and wideband multispectral optical spectrometers—play an essential part in the story.
Whether in the hospital or in the lab, optics technologies make possible rapid preliminary screening of potentially infected individuals, more accurate molecular diagnosis, reliable monitoring of disease progression and even, potentially, disinfection of contaminated surfaces. Our community developed these enabling technologies over the past several decades for applications ranging from telecommunications to machine and night vision. Now, they’re playing a life-saving role in the battle against SARS-CoV-2.
Safer screening
Early detection of infected patients—one of the primary challenges of the COVID-19 pandemic—is complicated by the wide variability in the disease’s symptoms. Monitoring for an increase in body temperature is the most commonly used preliminary screen. Under normal circumstances, direct body-cavity temperature measurements are the most accurate way to monitor a fever; however, given the pathogenicity of SARS-CoV-2, remote, non-contact options that employ infrared imaging cameras to simultaneously image and measure groups of individuals provide a significant safety advantage.
Many clinicians now rely on infrared-based thermometers for measuring forehead temperature. These imaging and spot-measurement thermometric devices provide medical personnel with a safer and useful non-contact patient screen. These thermometers are based on single detectors or arrays of MEMS-based microbolometers or semiconductor diode detectors—thermal sensors that are sensitive in the far-infrared spectral region (8 to 14 µm) and detect changes in the blackbody radiation intensities in persons with above-normal body temperatures.
Molecular diagnosis

Cepheid doctor’s office RT-PCR instrument. [Image: Courtesy of Cepheid]
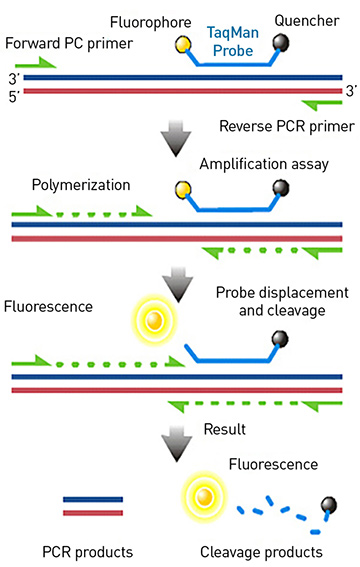
In “TaqMan” real-time polymerase chain reaction, a nucleic-acid probe molecule, tagged with a fluorescent molecule and an accompanying quencher, attaches to the stretch of DNA or RNA being copied. With each round of amplification, the fluorescent molecule is released into the buffer solution and separated from the quencher, allowing the amplification of the targeted genetic sequence—such as one from SARS-CoV-2—to be detected via fluorescence in real time. [Image: Wikimedia Commons]
If a patient presents with a fever or other symptoms typical of viral infection (sore throat, dry cough, muscle aches and fatigue), the next step is a molecular diagnostic test. This screen, based on a technique called real-time reverse transcription polymerase chain reaction (RT-PCR), uses sensitive spectroscopic methods to detect extremely small quantities of viral genetic material from a patient’s nasal or throat swab. And once again, optical technology is an essential component for disease detection.
The diagnostic procedure requires significant sample processing, beginning with a specimen collected from a patient. Real-time RT-PCR works by copying specific nucleic acid sequences within that sample, using probes—nucleic-acid primers—that selectively bind very specifically to the RNA sequences present in the SARS-CoV-2 virus. The probes are tagged with molecules of fluorescent dye.
Enzymes are then used to copy the nucleic-acid sequences bound to the probes. The sample is thermally cycled roughly 40 times between
37 °C and 95 °C. If the target nucleic-acid sequences are present, they are amplified twofold with each cycle.
It is optical technology that puts the “real time” in RT-PCR. As the amplification enzymes create the duplicate copies, the fluorescent molecules are released into the buffer solution. The overall fluorescence is measured in real time after each cycle, increasing as the number of amplicons increases for positive samples. By measuring the intensity buildup during the thermal cycling, the virus is detected and the amount of virus present (the viral load) can be estimated.
Real-time RT-PCR instruments employ narrowband visible laser diodes or LEDs as excitation sources and semiconductor diodes or photomultipliers with narrow band-pass optical filters for detection. These instruments are fully automated and can typically process 96 or 384 samples in parallel in less than an hour.
Real-time RT-PCR is one of the most sensitive and specific molecular-analysis techniques available today. This assay is crucial for tracking and controlling the spread of COVID-19. However, the overall sensitivity of the method may be limited by the efficiency of the sample collection and preparation process. The amount of virus present in the sampled tissue, which varies between individuals and as the disease progresses in each patient, may also be a limiting factor.
The false-negative rate of this approach is currently estimated at roughly 30%. Repeated testing can reduce this admittedly significant percentage, which is why many hospitals require two or three sequential negative real-time RT-PCR tests after a patient has recovered before that patient is classified as non-infectious.
Tracking disease progression
In addition to molecular diagnostics, imaging of the lungs of COVID-19 patients has also proved very sensitive for detecting SARS-CoV-2 infection using high-resolution computed tomography (CT) scans. Clinicians look for signs of lung damage as evidenced by “ground-glass” patterns in the lung tissue or fluid accumulation as signatures of pneumonia. Clinics in China have reported that this approach can detect a significant number of infected individuals that have negative RT-PCR readings—only, however, later in disease progression, once lung damage manifests.
If a patient is diagnosed with COVID-19, disease progression and respiratory function are determined using an oxygen-saturation meter, which measures the percentage of oxygenated hemoglobin in blood. As the disease progresses, breathing can become difficult, causing a reduction in oxygenated hemoglobin—if levels dip below certain thresholds, then supplementary oxygen or a ventilator may be warranted.
Oxygen-saturation devices use LEDs emitting at two different wavelengths, typically around 665 nm and 894 nm. The oxygen-saturation percentage is measured from the ratio of the absorption at these two wavelengths. These battery-powered devices fit comfortably on a finger or toe, providing real-time measurement of oxygen-saturation levels.
Measuring immunity: ELISA

96 sample well plate ELISA instrument. [Image: ©2020 Berthold Technologies. Used under permission. www.berthold.com]
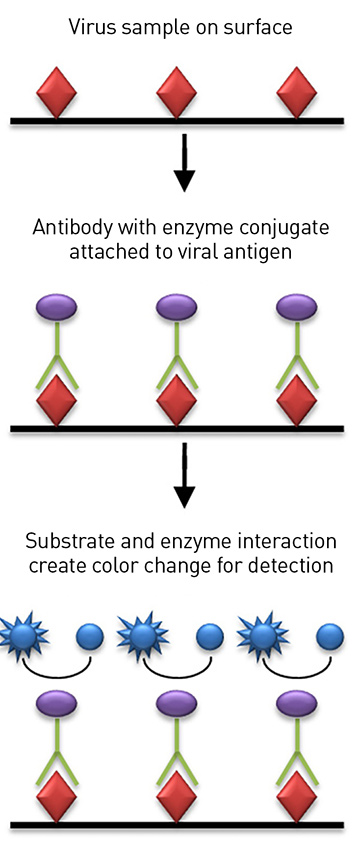
Schematic of ELISA, which measures the presence of specific antibodies in a COVID-19 patient’s sample. The technique relies on a colorimetric change in the sample generated by an enzyme attached to antibodies specific to SARS-CoV-2 virus. [Image: Cavitri/Wikimedia Commons, CC-BY 3.0]
Optical instruments are also used to test whether a person has been exposed to SARS-CoV-2 virus and has developed an immune response. These instruments—which can be automated to analyze hundreds to thousands of samples per day—use a technique called an Enzyme-Linked Immunosorbent Assay (ELISA) to measure the presence of antibodies specific to the SARS-CoV-2 virus in a patient’s blood-serum sample.
In a typical assay, an antigen found on the virus surface is immobilized on the bottom of a sample well, which is optically transparent. Antibodies in the serum sample are attached to an enzyme (typically horseradish peroxidase) and allowed to incubate on the surface containing the immobilized antigen. Any antibodies specific for the SARS-CoV-2 antigen bind to the target and become immobilized on the surface of the optical window. The unbound, nonspecific antibodies are washed off.
A solution containing the enzyme’s substrate with a colorimetric indicator is then added to the sample well, and the enzyme linked to the antibody reacts with the substrate, producing a color change in the sample. The enzyme reacts with multiple substrate molecules, thereby amplifying the signal. SARS-CoV-2 antibodies in the blood serum can then be detected and quantified, via multispectral imaging of the sample substrate’s fluorescence or absorption indicator.
This approach is used to measure the extent of the virus’ spread within a community, even after the pandemic has passed; to measure the duration of an individual’s immune response; and to investigate the efficacy of antiviral drug candidates and potential vaccines. Currently, medical workers who have recovered from COVID-19 and have a protective immune response to the virus are being identified using an ELISA. Once immunity is confirmed, these personnel safely resume working with infected patients—a common approach in pandemic medicine.
Toward vaccines and sterilization
Optical devices also form the core technology for the most common high-throughput gene-sequencing instruments. These typically use high-quantum-efficiency, very-high-resolution multispectral cameras to map the sequences of hundreds of millions of target DNA molecules simultaneously and can sequence the complete genome of the SARS-CoV-2 virus in just a few hours. Virus genetic sequences can vary with location, since the SARS-CoV-2 virus occasionally mutates during its replication phase. Infections in separate geographic regions can be compared, and the origins of infections traced, by comparing the specific mutations in samples taken from patients in different locations.
High-throughput sequencing of the virus genome also can determine the proteins in the virus and identify suitable targets for synthetic vaccines that will safely stimulate immune response. This technology has greatly improved over the past 20 years, largely due to the human genome project, and will be an essential tool for developing effective vaccines and antiviral drugs to combat the COVID-19 pandemic.
Prototype of an LED sterilization system being tested by Bolb Inc. [Image: Bolb Inc.]
Beyond the molecular-biology lab, optics is emerging as a weapon on another vital front: the sterilization of surfaces. Most viruses and bacteria are very sensitive to ultraviolet light, particularly in the UV-C spectral region (200–280 nm), which causes mutations in the RNA that is essential for viral replication. Recently, great progress has been made in the development of UV LEDs that emit in this region. LED arrays emitting hundreds of milliwatts have been developed with lifetimes of over 1000 hours and electrical efficiencies around 10%.
Arrays of these diodes can generate significant UV power levels to potentially decontaminate certain surfaces more efficiently than chemical reagents. Recent lab results indicate that exposure times of about 1 minute were sufficient to kill bacteria and viruses with a 1-W-average-power device located about 1 meter above a contaminated surface. Further testing on the efficacy of UV LEDs for decontaminating surfaces infected with SARS-CoV-2 virus is in progress.
Future needs
As global health faces this novel and deadly threat, laboratories around the world are using technologies developed by the optics and photonics community to help stem the spread and save lives. In the near future, as social distancing begins to slow the spread of COVID-19 disease, medical focus will shift to the early detection and isolation of COVID-19 recurrence in hot spots, which will present new challenges for diagnostic and decontamination technologies. These challenges represent new opportunities for optics and photonics technologies—with their advantages of low cost, high speed, sensitivity and specificity—to make major contributions to global health. OPN
Note: This article will also be published in the May 2020 issue of Optics & Photonics News.
2009 OSA President Thomas M. Baer (tmbaer@stanford.edu) is with Stanford University, USA. Christina E. Baer is with the University of Massachusetts Medical School, USA.
