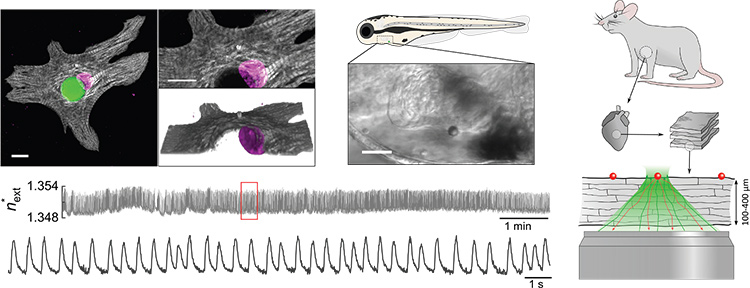
 Top left: Confocal microscopy shows a polymer microlaser (green) inside the cytosol of a neonatal cardiomyocyte, surrounded by the sarcomeric protein cTnT (grey) and the cell nucleus (magenta). Scale bars: 10 µm. Top center: A microlaser attached to the heart of a zebrafish. Scale bar: 50 µm. Bottom left: A reconstructed refractive-index profile of a continuously excited intracellular microlaser showing detailed contractility information on the single-cell level. Right: Illustration of the measurement of cardiac contractility in living myocardial slices up to a tissue thickness of 400 µm.
Top left: Confocal microscopy shows a polymer microlaser (green) inside the cytosol of a neonatal cardiomyocyte, surrounded by the sarcomeric protein cTnT (grey) and the cell nucleus (magenta). Scale bars: 10 µm. Top center: A microlaser attached to the heart of a zebrafish. Scale bar: 50 µm. Bottom left: A reconstructed refractive-index profile of a continuously excited intracellular microlaser showing detailed contractility information on the single-cell level. Right: Illustration of the measurement of cardiac contractility in living myocardial slices up to a tissue thickness of 400 µm.
The beating heart represents an exceptionally demanding environment for even the most advanced microscopy techniques, due to ultrafast tissue dynamics and severe light scattering by dense muscle fibers.1 However, characterizing the contraction of single cardiac cells is of fundamental importance for understanding heart disease, for improving heart transplants and for advancing cardiac stem cell therapies.2 To overcome the limitations facing cardiac sensing, we explored the integration of microscopic lasers as novel, ultra-sensitive and remarkably bright probes of heart tissue’s contractility.3
Previously, it had been shown that spherical whispering-gallery-mode microlasers can be actively internalized by a variety of biological cells, and can generate self-sustained laser emission from within those individual cells under remote optical pumping.4 We found that, due to the sensitivity of microlasers to local changes in refractive index, analysis of the emission spectra of microlasers placed in heart muscle cells (cardiomyocytes) allowed us to extract transient profiles of contractility in their immediate environment (see accompanying video). The results are quantitative and can be linked to the protein density of the contractile filaments (myofibrils) that form the muscle fibers. The spectral profile of each microlaser also provides a unique optical barcode to track individual heart cells over extended time periods.
To explore the translational perspective of microlaser-based contractility probes, optical cardiac sensing was performed in the hearts of live zebrafish. Detailed single-cell contractility profiles were recorded, demonstrating that the technique is not affected by the fast movements of the heart itself.
Finally, we explored the ability to detect these profiles deep inside scattering tissue by integrating microlasers into living myocardial slices. Currently available multiphoton microscopes fail to resolve contractions of single cells in slices beyond a depth of only 100 µm.1,3 In contrast, microlaser sensing in cardiac slices offered much deeper penetration, allowing us to reliably detect contractility at tissue depths of up to 400 µm without the need for multiphoton excitation. In combination with newly developed nanolasers,5 this technique will stimulate novel translational approaches by reliably accessing the functional properties of transplanted cells and engineered cardiac tissue noninvasively—and deep within the beating heart.
Researchers
Marcel Schubert and Malte Gather, University of St. Andrews, St. Andrews, U.K.
References
1. A.D. Aguirre et al. Proc. Natl. Acad. Sci. U.S.A. 111, 11257 (2014).
2. E. Tzahor and K. Poss. Science 356, 1035 (2017).
3. M. Schubert et al. Nat. Photon. 14, 452 (2020).
4. M. Schubert et al. Nano Lett. 15, 5647 (2015).
5. A.H. Fikouras et al. Nat. Commun. 9, 4817 (2018).
