 [Getty Images]
[Getty Images]
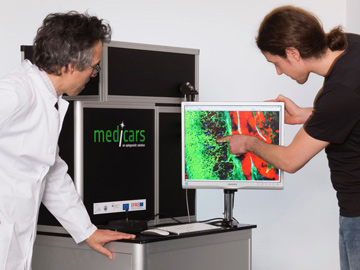 A compact experimental setup for multimodal nonlinear microscopy in clinical settings. [Leibniz-IPHT, S. Döring]
A compact experimental setup for multimodal nonlinear microscopy in clinical settings. [Leibniz-IPHT, S. Döring]
Aging populations, exposure to environmental contamination in developing countries, and other factors are driving significant increases in cancer worldwide. The World Health Organization estimates that globally, the number of cancer cases will reach 21.6 million by 2030, nearly double the 14 million patients in 2012. In Germany alone, malignant tumors are responsible for a quarter of all deaths.
That kind of growth will raise new challenges—in the early detection of malignant lesions, which can significantly improve patient outcomes; in precise determination of tumor boundaries for surgery, which can avoid incomplete resections and cancer recurrence; and in rapid determination of tumor type and grade, which can cut back on the number of surgeries and save precious time in the operating room. Unfortunately, none of these challenges are adequately addressed today. In large part, that’s because reliably diagnosing and characterizing cancer, especially for surgical interventions, is a complex process involving a variety of imaging methods and, ultimately, histopathology, the time-consuming, microscopic analysis of stained tissue biopsies and cell material.
To improve patient outcomes—and also to reduce treatment costs, a burden in both developing nations and developed countries with aging populations—will require new techniques to provide diagnostically relevant information, quickly and reliably. And we believe that optics could play a crucial role. Indeed, the maturing of photonic technologies has created an opportunity to develop an efficient, noninvasive, all-optical biopsy that could help meet the challenges of clinical pathology in an era of rising cancer incidence. In this feature, we take a closer look.
The maturing of photonic technologies has created an opportunity to develop an efficient, noninvasive, all-optical biopsy to help meet the challenges of clinical pathology.
One name, many diseases
Before examining how photonic technologies could aid in cancer diagnosis, it’s worth briefly considering why malignant tumors are so difficult to fight. Fundamentally, the reason is that cancer is not one disease, but many.
While virus-driven forms of cancer do exist, most cancers, unlike infectious diseases, are not caused by viruses, bacteria or abnormal infectious protein fragments (prions), but are initiated by accumulated genetic mutations within dividing cells. Typically, cells either correct for such DNA damage or, if the damage can’t be repaired, initiate “programmed cell death,” or apoptosis. Many types of cancer thus work through mutations of so-called oncogenes, which block these important cellular repair or housecleaning mechanisms. Several hundred such oncogenes are known to date.
Every tumor thus possesses a particular combination of mutations that suppress specific cellular proteins or processes (especially the immune response). As a result, a variety of drugs or treatment strategies can be used, including some, such as radiotherapy and chemotherapy, that focus on the destruction of fast-growing cancer cells, and emerging therapies that work by stimulating the body’s immune response. Because the particular pattern of mutations within cells differs so much from patient to patient, these therapies must be individually adjusted to optimize tumor suppression and reduce side effects. Indeed, cancer’s highly diverse nature puts a premium on accurately characterizing tumor stage and grade, to identify patients who can benefit from emerging multimodal therapies combining surgery, radiotherapy, chemotherapy, antibody therapy and immunotherapy.
 [Getty Images]
[Getty Images]
Case study: Head and neck cancers
Can a multicontrast optical approach improve our ability to get a clinical view of cancer? To explore the question, we look at a specific type of cancer—head and neck squamous cell carcinoma (HNSCC)—and the current state of the art for tumor diagnosis and characterization.
HNSCC is the sixth most common cancer worldwide and the third most common in the developing world. As its name implies, the disease involves the squamous cells that line the moist, mucosal surfaces inside the head and neck. HNSCC’s incidence correlates with tobacco use, with alcohol consumption and, in some subsites, with human papillomavirus (HPV) infection. While early detection significantly improves overall survival rates, early-stage HNSCC lesions are visually quite similar to the inflammations and other lesions common in the oral cavities of heavy smokers and drinkers. Patients thus often come to the clinic with several suspicious lesions.
The current gold standard in cancer diagnostics involves removing a tissue sample for a biopsy and histopathologic examination. The painstaking, time-consuming process begins with excision of the tissue—which is uncomfortable to the patient, and can be subject to sampling bias—and moves onto formalin fixation (FFPE), paraffin embedding, tissue sectioning, staining with an agent such as hematoxylin-eosin (H&E), and evaluation by a trained pathologist.
If histopathology results in diagnosis of a head and neck tumor, surgical therapy is usually performed. But whereas in some other cancers, tissue can be generously excised around the tumor to ensure complete removal, this would be far too dangerous for head and neck tumors, as doing so could significantly reduce function, especially when the tumor lies in close proximity to important structures like the vocal folds or the epiglottis. Thus, both function needs to be preserved and the tumor needs to be removed as completely as possible.
At present, the procedure for ensuring tumor-free margins in HNSCC surgeries involves frozen-section histopathology during the operation itself. Tissue is excised and shock-frozen in liquid nitrogen, after which a trained pathologist investigates H&E-stained sections of the tumor margin—a process that can take 20 minutes or longer. Meanwhile, progress in the surgery halts until results from the pathologist either confirm tumor-free margins or identify residual tumor cells, in which case surgical resection continues.
Tools for an all-optical biopsy
Optical coherence tomography
Penetration depth: >1 mm
Spatial resolution: 1 µm
Speed (voxel/s or spectra/s): ~109
Basis of contrast generation: Δn
Second-harmonic generation
Penetration depth: ~1 mm
Spatial resolution: <1 µm
Speed (voxel/s or spectra/s): ~106–107
Basis of contrast generation: χ(2) (e.g., for collagen)
Third-harmonic generation
Penetration depth: ~1 mm
Spatial resolution: <1 µm
Speed (voxel/s or spectra/s): ~106–107
Basis of contrast generation: Δn (nonlinear)
Two- and three-photon excited fluorescence
Penetration depth: ~1 mm
Spatial resolution: <1 µm
Speed (voxel/s or spectra/s):~106–107
Basis of contrast generation: Autofluorescence
[Elastin, FAD, Keratin, NAD(P)H]
Photoacoustic tomography
Penetration depth: ~3-5 cm
Spatial resolution: >100 µm
Speed (voxel/s or spectra/s): ~106–107
Basis of contrast generation: Absorption
Diffuse optical tomography
Penetration depth: ~3-5 cm
Spatial resolution: >100 µm
Speed (voxel/s or spectra/s): ~106–107
Basis of contrast generation: Absorption
Hyperspectral imaging
Penetration depth: ~mm
Spatial resolution: ~50 µm
Speed (voxel/s or spectra/s): ~100000
Basis of contrast generation: Absorption
Raman
Penetration depth: ~few 100 µm
Spatial resolution: <1 µm
Speed (voxel/s or spectra/s): ~10
Basis of contrast generation: Inelastic scattering
(molecular vibrations)
Δn = refractive-index contrast
χ(2) = second-order nonlinear optical susceptibility
Since frozen-section histopathology, performed during surgery, is less precise than standard, painstaking FFPE histopathology, some 10 percent of results from frozen-section analysis do not agree with the final histopathological examination. Such cases are handled with follow-up surgeries in combination with radiotherapy—a protocol that severely reduces not only the patient’s prognosis but also their overall quality of life.
A suite of optical tools
Particularly given the current state of the art, it is not difficult to see how less-invasive optical imaging methods could significantly benefit both clinicians and patients with HNSCC. For early tumor detection and diagnosis, high-resolution imaging could enable determination of a lesion’s status without tissue removal, and allow investigation of all suspicious lesions rather than a small sample potentially vulnerable to sampling bias. In the operating room, imaging methods capable of quickly detecting the tumor’s extent would significantly improve surgical guidance and reduce operation times; if those imaging methods also enabled tumor typing, additional time could be saved and the treatment strategy optimized.
In the past decade, the use of optical methods in medical diagnostics has rapidly increased, both driven by advances in biophotonics and further moving the field ahead. Particularly within the past few years, biophotonics has witnessed the development of methods and technologies that potentially could meet the challenges of clinical pathology outlined above, through the implementation of a genuine optical molecular pathology. We believe that this perspective, drawing together a variety of optical imaging modalities toward a common purpose, could enable a true noninvasive, all-optical biopsy of tissue volumes in the near future.
What would such a multimodal photonic-imaging approach need to achieve? To provide a superior alternative for current methods to determine tumor type—currently accomplished via genomics and DNA analysis, or through immunohistochemistry, involving fluorescently labeled antibodies—and for the detection of tumor extent and characteristics currently enabled by histopathology, in vivo imaging of tumors would need to provide four things:
-
high resolution, to visualize subcellular details;
-
label-free and molecule-specific detection, to visualize subtle changes in chemical composition during initiation of carcinogenesis;
-
high speed, enabling real-time imaging; and
- high depth penetration, on the order of millimeters, in 3-D, to investigate the extent of a lesion noninvasively.
Clearly, no single optical method can meet all of these requirements. The large data volumes of high-resolution techniques mean that they usually cannot proceed at high speed; similarly, molecular contrast typically requires spectral information (such as Raman or IR-spectroscopy), which also slows down data acquisition. Conversely, the fastest methods, such as optical coherence tomography (OCT), commonly provide limited information and spatial resolution when carried out at high depth penetration.
But putting together a suite of available tools, we believe, can accomplish all of the requirements, and point the way toward an all-optical biopsy. In the rest of this article, we look at the important characteristics of these existing imaging modalities, and then focus on one specific strategy for an optical biopsy, multimodal nonlinear imaging.
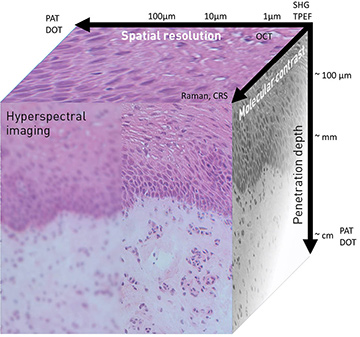 Spatial resolution, depth penetration and level of molecular information for a number of common photonic imaging methods. Photoacoustic tomography (PAT) and diffuse optical tomography (DOT), for example, provide high penetration (~cm) but rather low spatial resolution (>100 μm). [Courtesy of the authors]
Spatial resolution, depth penetration and level of molecular information for a number of common photonic imaging methods. Photoacoustic tomography (PAT) and diffuse optical tomography (DOT), for example, provide high penetration (~cm) but rather low spatial resolution (>100 μm). [Courtesy of the authors]
Methods and strategies
A variety of photonic imaging methods have matured in recent years to be applied in medical imaging, with a few (such as OCT in ophthalmology and dermatology) even entering routine diagnostics. The photonic methods include established techniques like wide-field microscopy with specific excitation wavelengths or using medically approved vital stains like 5-ALA or indocyanine green; special spectral ranges, like narrowband imaging; or broadband illumination and hyperspectral imaging, currently under active investigation.
Hyperspectral imaging generates spectrally resolved wide-field image data that enables determination of parameters such as hemoglobin content and tissue oxygenation, both superficially and at depth, as well as water and lipid content. More sophisticated imaging methods include confocal or nonlinear laser scanning microscopic imaging using different contrast mechanisms, such as Raman scattering, fluorescence imaging, fluorescence lifetime imaging (FLIM), second-harmonic generation (SHG) and third-harmonic generation (THG).
These imaging modalities can be distinguished by the method used for image generation, and by the underlying contrast mechanisms. The latter can be highly molecule specific in case of infrared (IR) absorption and Raman scattering; can visualize native electronic chromophores such as hemoglobin, NADP(H), flavine, elastin and cythochrome by absorption (photoacoustic imaging) or emission (autofluorescence imaging); or can highlight refractive-index changes (OCT).
How do these various techniques stack up in terms of the four key imaging parameters outlined above?
Penetration depth. The penetration depth in tissue is closely related to photon transport. Photons travel ballistically at low depths, but at depths significantly larger than the mean free path they become strongly scattered, and their travel is diffusive. The deepest penetration depth is currently achieved by three-photon fluorescence in the brain in on the order of 1.5 mm, while in the diffusion limit, photoacoustic or diffuse optical tomography can reach depths of up to several centimeters.
Resolution. High resolution of subcellular details can be realized only for ballistically travelling photons, using techniques such as confocal or nonlinear laser scanning microscopy.
Molecular specificity. The molecular contrast is highest if spectral data are acquired, such as in hyperspectral imaging or spontaneous or coherent Raman spectroscopy. These techniques allow visualization of the spatial distribution of molecular markers like proteins, lipids or DNA, whereas methods such as OCT detect only changes in refractive index, which cannot be correlated with a specific molecular structure. Acquisition of spectral information is time consuming, however, making such methods generally slower than techniques measuring just a single parameter per pixel, such as OCT.
Speed. OCT is currently the fastest method in terms of volume elements (voxels) imaged per second. With sophisticated Fourier-domain mode-locked laser sources, the acquisition of up to 4.5 GVoxel of data per second has been demonstrated in OCT, allowing for real-time 3-D imaging of dynamic processes.
As already suggested, to fulfill the above-mentioned four clinical requirements, an all-optical biopsy will not rely on a single imaging modality, but on the clever combination of several imaging approaches. One can think of two end-member strategies for combining these diverse methods. On one end, imaging modalities with similar acquisition speed and resolution can be combined—for example, nonlinear imaging methods like two-photon excited fluorescence, SHG and coherent Raman scattering, all of which are efficiently excited by the same laser source and can be detected in parallel.
On the other end, imaging modalities of different imaging speed and tissue penetration can be synergistically combined, so that a fast but chemically less specific method provides an overview on the tissue volume, while a slower, molecule-specific second method is used to classify tissues detected by the faster modality in suspicious areas. One such approach would be to combine OCT or FLIM with Raman.
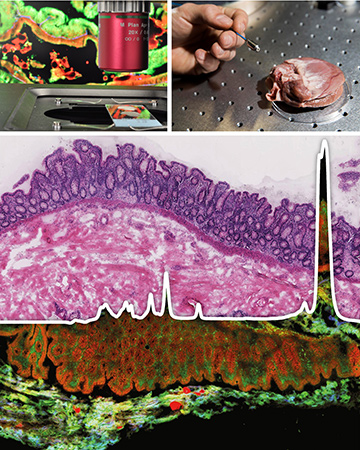 Top: Multimodal nonlinear imaging can be done either in thin tissue sections ex vivo (left) or in vivo using a compact fiber optic probe (right). Middle and bottom: The multimodal nonlinear microscopic false-color image contains spectral information on the spatial distribution of a number of biomolecules, which can be used to calculate an image resembling a stained histopathological section (a so-called virtual or pseudo-H&E image). [Lab images courtesy of the authors; photo in upper right from Leibniz-IPHT, S. Döring]
Top: Multimodal nonlinear imaging can be done either in thin tissue sections ex vivo (left) or in vivo using a compact fiber optic probe (right). Middle and bottom: The multimodal nonlinear microscopic false-color image contains spectral information on the spatial distribution of a number of biomolecules, which can be used to calculate an image resembling a stained histopathological section (a so-called virtual or pseudo-H&E image). [Lab images courtesy of the authors; photo in upper right from Leibniz-IPHT, S. Döring]
Multimodal nonlinear imaging
In the next few paragraphs, we explore one possibility for an all-optical biopsy under the first strategy—combining coherent anti-Stokes Raman scattering (CARS), second-harmonic generation (SHG) and two-photon excited fluorescence (TPEF) in a multimodal nonlinear imaging approach. This approach demonstrates in particular the importance of sophisticated image analysis to derive “virtual” histologic stains useful in the clinic. (For a bit more background on these methods, see “Multimodal Nonlinear Optical Microscopy,” Optics & Photonics News, March 2014, p. 42.)
Multimodal nonlinear imaging for an optical biopsy begins with the investigation of a lesion—either of a thin tissue section via laser scanning microscopy, or in vivo using a fiber probe. In the scan, a special, ps-pulse laser source emits two wavelengths simultaneously, with a frequency difference that matches a Raman active vibration of the molecules of interest. Commonly, a frequency difference of 2850 cm–1 is used to image lipid distributions and to visualize membranes using CARS. SHG, meanwhile, is sensitive to noncentrosymmetric protein assemblies, particularly collagen fibers of the connective tissue, while TPEF visualizes the distribution of autofluorescence of molecular species such as fluorescent proteins keratin and elastin or co-factors like NAD(P)H or FAD. Thus, a single laser source can provide data across a variety of factors of interest, via three complementary nonlinear optical techniques.
The combined image data form a false-color image that can be transferred to a histologic-stain-equivalent image, thereby mimicking a white-light image of the section stained with H&E. This allows the same section to be analyzed using different imaging techniques—for example, when a suspicious region needs to be examined in more detail—since the tissue itself has not been modified. Also, because multimodal nonlinear microscopy can be applied in vivo using a compact fiber optic probe, it can investigate multiple suspicious lesions noninvasively. And this optical approach can be used intraoperatively for surgical guidance, to evaluate the status of the resection margin when performing a tumor surgery, so that excision is continued until the resection margins are tumor free.
For head and neck cancers in particular this strategy is highly promising, as surgery using CO2 lasers is already routine in head and neck tumor surgery. Precise, real-time optical tools for surgical guidance offering diffraction limited spatial resolution of subcellular structures could thus significantly improve the precision of laser surgery, better preserve functional structures and reduce side effects, while at the same time reducing the number of false negatives on tumor margins.
A better outlook
As we have tried to show here, synergistically combining optical imaging modalities could allow clinicians to thoroughly investigate tissue morphologies and molecular structures without using exogenous markers—a highly promising combination for in vivo tumor diagnostics and intraoperative surgical guidance. Many tools are under development, but only a few routine clinical applications have emerged thus far owing to complex equipment and the difference of this approach from existing methods.
The tools of the all-optical biopsy must still be validated against standard diagnostics in clinical trials, to prove their potential to overcome the limitations and drawbacks of standard histopathology. Significant potential progress could lie in implementing fast and complex algorithms for automated data processing and interpretation, especially using artificial-intelligence-based methods, like machine learning or deep learning. Furthermore, while to date a couple of methods have been readily combined, an even larger set of optical techniques will likely need to be put together to meet all clinical requirements in terms of speed, resolution, penetration depth and molecular specificity. For instance, it is likely that the method of multimodal nonlinear microscopy outlined above will be combined with pointwise Raman spectroscopy and OCT for large-volume screening (on the order of mm3 ), while wide-field methods like hyperspectral imaging complement the set of tools for scanning cm-sized tissue areas for suspicious zones.
Such a multimodal approach, we believe, could dramatically improve the diagnostics of tumors and the precision of surgical therapies—leading to better outcomes for physicians and patients alike.
Financial support of the European Union, the Thüringer Ministerium für Wirtschaft, Wissenschaft und Digitale Gesellschaft, the Thüringer Aufbaubank, the Federal Ministry of Education and Research, Germany (BMBF), the German Science Foundation, the Fonds der Chemischen Industrie and the Carl-Zeiss Foundation is gratefully acknowledged.
Tobias Meyer is with the Leibniz Institute of Photonic Technology e.V, Jena, Germany. Michael Schmitt is with the Institute of Physical Chemistry & Abbe Center of Photonics, Friedrich-Schiller University, Jena, Germany. Orlando Guntinas-Lichius is with the Department of Otorhinolaryngology, University Hospital, Jena, Germany. Jürgen Popp is with the Leibniz Institute of Photonic Technology e.V and the Institute of Physical Chemistry & Abbe Center of Photonics, Friedrich-Schiller University, Jena, Germany.
References and Resources
-
N. Vogler et al. “Multimodal imaging spectroscopy of tissue,” Ann. Rev. Anal. Chem. 8, 359 (2015).
-
T.W. Bocklitz et al. “Pseudo-HE images derived from CARS/TPEF/SHG multimodal imaging in combination with Raman-spectroscopy as a pathological screening tool,” BMC Cancer 16, 534 (2016).
-
O. Chernavskaia et al. “Beyond endoscopic assessment in inflammatory bowel disease: Real-time histology of disease activity by non-linear multimodal imaging,” Sci. Rep. 6, 29239 (2016).
-
A. Lukic et al. “Endoscopic fiber probe for nonlinear spectroscopic imaging,” Optica 4, 496 (2017).
