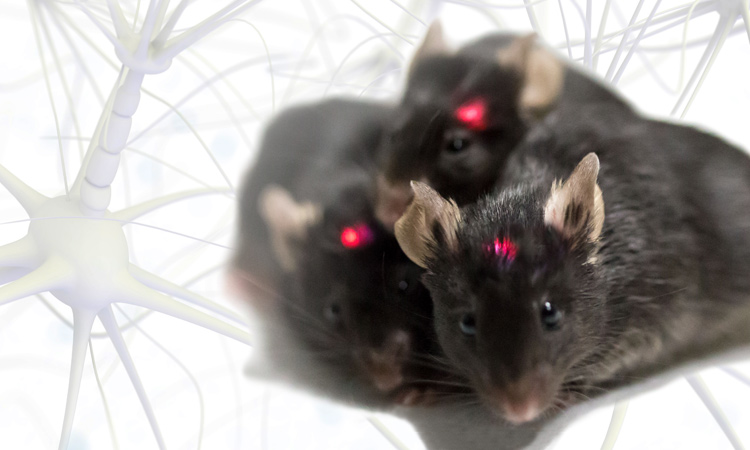
 Wireless optogenetics tools like these tiny implants in live mice are enabling scientists to map the stimulation of certain neurons of the brain to specific responses. [J. Rogers/Northwestern Univ.]
Wireless optogenetics tools like these tiny implants in live mice are enabling scientists to map the stimulation of certain neurons of the brain to specific responses. [J. Rogers/Northwestern Univ.]
Two thousand years ago, ancient Egyptians knew that the electrical shocks of torpedo fish, applied to the body, could offer pain relief. Two hundred years ago, physicians understood that electrical stimulation of a frog’s spine could control muscle contraction. Today, electrical therapy underlies many treatments, from pacemakers to pain control.
But neuroscience has long awaited a more precise tool for controlling specific types of neurons. Electrical stimulation (e-stim) approaches stimulate a large area without precise spatial control, and can’t distinguish between different cell types. E-stim also can induce unwanted thermal heating of tissue and other parasitic effects. Drugs are a powerful alternative to trigger a similar neuronal sequence, but they are imprecise for use in the brain and much slower than the brain’s natural mechanisms.
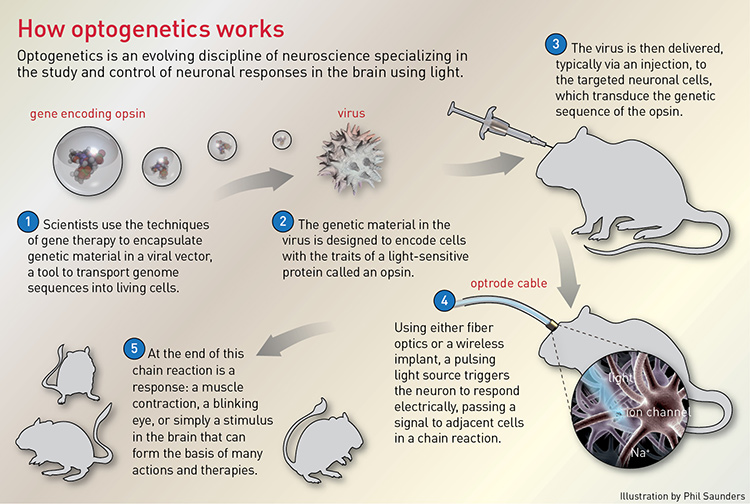
In the 1980s, Nobel laureate Francis Crick suggested that light might have the right stuff for controlling neurons of different types, thanks to its ability to provide timed pulses with millisecond temporal precision. However, light penetrates only a few hundred microns into the surface of the brain, scattering without affecting signaling. The challenge became: How can we get the brain to respond to light? This question gave rise to the field of optogenetics, and the emerging idea of replacing electrical stimulation with a combination of optics and genetic modification.
In just over a decade, the discovery of numerous opsins with different specializations has allowed scientists and engineers to make rapid progress in mapping brain activity.
Opsins and dollars
In the early 2000s, researchers Edward Boyden and Karl Deisseroth at Stanford University, USA, approached the problem by attempting to genetically encode brain cells with photosensitive properties. In 2004, they successfully confirmed that the opsin called channelrhodopsin, a microbial photosensitive protein derived from algae, could be transduced into neurons to control their electrical signaling, turning them on by opening neuronal ion channels in response to pulses of blue light. Other opsins followed, including those that suppress neuronal activity; for example, halorhodopsin can silence the neurons in the hypothalamus, inducing sleep in living mice.
In just over a decade, the discovery of numerous opsins with different specializations has allowed scientists and engineers to make rapid progress in mapping brain activity, motivated by the hope of solving intractable neurological conditions. And it doesn’t hurt that investment in neuroscience research has grown at the same time.
In 2013, the Obama administration announced a collaborative public-private effort, the Brain Research through Advancing Innovative Neurotechnologies (BRAIN) Initiative. The ten-year initiative aims to support the development and application of technology that will help in understanding the human brain and treat brain disorders such as Alzheimer’s disease, traumatic brain injury, epilepsy and autism. In October 2017, the U.S. National Institutes of Health (NIH) announced 110 new awards totaling US$169 million, bringing the total funding of the BRAIN program for 2017 to US$260 million. The 21st Century Cures Act, passed in December 2016, earmarked another $1.6 billion to the BRAIN Initiative over 10 years.
That may sound like a lot. But NIH estimates that the total funding needed to map the human brain in action over the course of a decade is around $4.5 billion.
Today, Boyden’s goal is to be able to map the brain and control it with enough precision to treat all brain disorders and simulate thoughts and feelings in a computer. Although light is inherently easier to control and aim when directed into tissue, it still scatters and diminishes over distance, reducing its effect. Boyden, now associate professor of biological engineering and brain and cognitive sciences at the MIT Media Lab and McGovern Institute (Cambridge, Mass., USA), and colleagues including coauthor Valentina Emiliani (Paris-Descartes, Paris, France) recently published results of a new optogenetic technique that addresses this issue. The method uses a new high-efficacy opsin and a two-photon holographic stimulation technique to illuminate single cells with unprecedented precise spatial and temporal control. The researchers were able to narrowly direct light to stimulate individual neurons in brain slices and to measure the response from connected, adjacent cells. The research sets the stage for more precise, millisecond-scale mapping of the connections in the brain in living animals.
 An ultraminiature, implantable, wireless device platform for optogenetics, developed by John Rogers and colleagues, measures 10 mm in diameter (smaller than a dime) and features a flexible micro-scale inorganic LED (µ-ILED) that fits through the eye of a needle (right), and that’s monolithically integrated into the end of an injectable silicon probe. [J. Rogers/Northwestern University]
An ultraminiature, implantable, wireless device platform for optogenetics, developed by John Rogers and colleagues, measures 10 mm in diameter (smaller than a dime) and features a flexible micro-scale inorganic LED (µ-ILED) that fits through the eye of a needle (right), and that’s monolithically integrated into the end of an injectable silicon probe. [J. Rogers/Northwestern University]
Wireless optogenetics
The most common type of optical system now used to map brain activity in mice involves a fiber optic cable attached to the mouse’s head to deliver pulses of light from an external light source. That has some obvious limitations: the size of the cable creates damage during insertion and irritation as the animal moves, and the subject must remain physically tethered to the light source, which limits social interaction and normal behavior.
The field is shifting with the advance of wireless optogenetics systems. In 2013, while a professor of materials science at the University of Illinois, Urbana-Champaign (USA), John Rogers, along with colleagues at Northwestern University (Evanston, Ill., USA), built the first wireless optogenetics system in which a micron-scale inorganic LED (µ-ILED) is injected directly into the mouse’s brain and powered wirelessly via radio antenna. The µ-ILEDs, fabricated with custom lithographic techniques borrowed from the integrated-circuit industry, consist of an active semiconductor material on a thin compliant polymeric structure that also serves as mechanical support.
The µ-ILEDs are the size of a single neuron, many orders of magnitude smaller than commercially available LEDs. The systems are ultralight, battery free, and allow wireless power transfer and control with the brain-injected µ-ILEDs. The custom wireless µ-ILED platforms were in such demand from neuroscientists that Rogers set up a small company, Neurolux (Champaign, Ill., USA), to respond to the requests. “Rather than just publish our results,” said Rogers, now Simpson/Querrey professor of materials science, engineering and surgery at Northwestern, “we wanted to follow up and make those technologies broadly available so they can have a more lasting impact on the field.”
Still an early-stage company, Neurolux is refining the µ-ILEDs and system components to be manufacturable for industry. While most optogenetic activity occurs between the red and blue parts of the spectrum, the Neurolux LEDs cover every color in the visible spectrum and into the UV. Researchers can select the LED wavelength based on the desired response: red light generally inhibits a response in the brain, blue light stimulates one, and green can do either depending on the targeted opsin.
The µ-ILED implant design is now single-use and disposable, and is mounted subdermally so that the implant is not noticeable in the animal. That allows mice to exhibit normal behavior, such as social interaction or movement through tunnels or on wheels. “The mice can do all the things that would otherwise be impossible with standard fiber optic setups,” said Rogers. Further development has enabled layering of the thin polymeric shank of the wireless implants with integrated microfluidic systems for pharmacological delivery, a temperature sensor, micro-photodetectors and a microelectrode.
In October 2017, Neurolux received a Phase I Small Business Technology Transfer (STTR) grant from NIH to further develop the company’s wireless optogenetic system for neural stimulation and localized, user-programmable pharmacological delivery. According to Rogers, in spite of the hurdles, many people believe that therapeutic optogenetics methods will ultimately be applied to humans. “Optogenetic modifications in mice have shown no adverse impacts to their health,” said Rogers, “so it’s conceivable that these approaches could one day be approved for safe use in humans.”
Self-tracking wireless
In 2015, Ada Poon, a Stanford associate professor of electrical engineering, led a multidisciplinary team taking another approach with wireless optogenetics: a self-tracking, wireless, fully implantable LED device for untethered optogenetic control throughout the nervous system in normally behaving mice. Like Rogers’ device, the entire stimulator, including receiving coil, circuit and LED, measures only 10 to 25 mm3 and weighs 20 to 50 mg, depending on the target neural structure. (For comparison, a mouse’s head weighs approximately 2 g.) The device can be implanted invisibly not just in the brain but in peripheral locations such as limbs or spinal cord. This expands its use beyond brain stimulation to experiments in drug testing and pain management.
 A resonant cavity (top) powers the wireless optogenetic platform developed by Ada Poon and colleagues at Stanford, using commercially available components. Wirelessly powered internal implants (inset, bottom) enable the mouse to move freely and behave normally while receiving optogenetic stimulation of peripheral nerve endings in the hind paw. [A. Poon/Stanford University]
A resonant cavity (top) powers the wireless optogenetic platform developed by Ada Poon and colleagues at Stanford, using commercially available components. Wirelessly powered internal implants (inset, bottom) enable the mouse to move freely and behave normally while receiving optogenetic stimulation of peripheral nerve endings in the hind paw. [A. Poon/Stanford University]
The system enables self-tracking using a microwave resonant cavity, a hollow metal structure with a honeycomb top plate on which the mouse stands. When excited by an RF source at a specific resonance frequency, the cavity confines electromagnetic fields inside it. The honeycomb metal plate acts as an energy transfer mechanism, so that everywhere a mouse steps on the honeycomb, the energy flows through the mouse’s body to reach the tiny receiving coil in the implanted LED device. The subcutaneous implant activates targeted clusters of neurons, which allows self-tracking of optogenetically stimulated mice safely and efficiently.
Poon hopes to use the system to learn where memories are stored in the brain, and thus to directly inform clinical studies. For example, a specific cluster of neurons in a mouse’s brain might identify the specific memory of a painful shock, which is observed because the mouse freezes in a defensive crouch.
“We hope that by revealing the stimulation patterns that cause Alzheimer’s mice to recall a shock, we’ll help scientists discover similar patterns that cause people with Alzheimer’s to remember other useful things, like the events of their lives or the faces of the people they love,” said Poon. “As optogenetic technology gets better and makes delicate brain research easier, I believe it can be a guiding light for neuroscience.”
“As optogenetic technology gets better and makes delicate brain research easier, I believe it can be a guiding light for neuroscience.”
—Ada Poon, Stanford University
But clinical solutions might be harder to obtain. Poon believes that optogenetics may never be a practical therapy for human brain diseases such as Parkinson’s or Alzheimer’s. Also an investigator affiliated with the Chan Zuckerberg Biohub (founded by Mark Zuckerberg and Priscilla Chan to help cure, prevent or manage all diseases within their children’s lifetime), Poon thinks that no single technology, like e-stim or optogenetics, will win at the outset. Instead, she believes, optogenetics will prove more useful in helping scientists understand the mechanics of the brain to inform new techniques that do not require genetic modification in humans.
Stepping up to primates
Until recently, optogenetic experiments have been largely restricted to rodents, which can provide only limited insight into the human brain. The next logical step in complexity is to study the brain function of nonhuman primates (NHPs), which have brains closer to the human brain than any other experimentally accessible species. Optogenetics in monkeys is a very new field, dawning in just the last decade.
Parkinson’s disease is a progressive movement disorder of the nervous system estimated to affect more than 10 million people worldwide. Parkinson’s is thought to derive from gradual cell death in the substantia nigra part of the basal ganglia in the brain, causing a shortage of the neurotransmitter dopamine, which is required to control voluntary and involuntary movements. Adriana Galvan, an assistant professor of neurology at Emory University (Atlanta, Ga., USA), uses optogenetics to explore the brain circuits related to movement planning and execution in normal rhesus macaques versus Parkinsonian macaques.
Galvan transitioned from rodents to NHP brain studies as a postdoctoral researcher in 2010. “It was not clear, back then, whether the technique that had worked so wonderfully in rodents would be useful in primates,” said Galvan. Even once that potential was established, many challenges remained. For one thing, it’s more difficult to deliver enough light to a large number of neurons to modulate behavior in larger animals. The area stimulated in monkeys using the same light sources and optical fiber used in rodents was too small. Researchers addressed this delivery problem by tapering the end of the optical fiber to illuminate a larger brain area, and by developing opsins with improved light sensitivity.
Another challenge was limiting the selective expression of opsins to one particular type of neuron. One cubic centimeter of your brain contains approximately 100,000 neurons with perhaps a billion connections between them. Scientists might be interested in studying connections between neurons of type A that project to brain region X, but must at the same time avoid stimulating type B neurons that project to region Y. Ideally, the opsins to be expressed would only be in type A neurons and not B.
Galvan and colleagues addressed this problem in 2016, using a trick called “terminal targeting.” They injected the virus carrying the genetic sequence for the opsin and let it be picked up by both A and B type neurons. Then, they delivered light via optical fiber only to brain region X, which then illuminated the terminals of A neurons. This way, the optogenetic stimulation affects only A–X connections, not B–Y.
Many unanswered questions remain about Parkinson’s, casting doubt on whether a therapeutic application of optogenetics is likely in the near future. However, Galvan hopes to use optogenetics to further explore brain circuits, map connections, and refine techniques to understand how stimulation of different brain regions affects movement in normal monkeys compared to Parkinsonian monkeys. “The answers to these questions are very important to guide novel therapies,” said Galvan. “And we may see optogenetics-inspired techniques in clinical use within 10 years.”
 An optrode (an optical fiber glued to an electrode for measuring electrical activity of illuminated neurons) features a tapered electrode ending (top) for minimal tissue damage in larger brains of nonhuman primates during optogenetic stimulation. When switched on (bottom), the optrode emits a narrower, targeted cone of blue light. [A. Galvan/Emory University]
An optrode (an optical fiber glued to an electrode for measuring electrical activity of illuminated neurons) features a tapered electrode ending (top) for minimal tissue damage in larger brains of nonhuman primates during optogenetic stimulation. When switched on (bottom), the optrode emits a narrower, targeted cone of blue light. [A. Galvan/Emory University]
Continuing research in primate optogenetics may also inspire practical engineering innovations that could be applied to humans. For example, optical fibers used in current laser thermal ablation procedures on the human brain, such as in brain tumors or epileptic therapy, measure approximately 600 µm in diameter, whereas in Galvan’s optogenetics experiments on primates, the fiber diameter is smaller, about 200 to 400 µm. Tapering the fiber to increase light delivery could enable smaller probes in human surgery that could translate into less tissue damage.
Human trials
While hundreds of recent studies have examined how optogenetics can map behavior and responses in the brains of mice and NHPs, scientists have yet to use such optogenetic mapping in the human brain. Researchers are, however, pursuing optogenetic techniques as a promising therapeutic pathway to treat physiological systems beyond the brain, such as the cardiovascular system, vision and the spinal cord. In fact, 2018 will see several commercial entities involved in early-stage clinical trials of optogenetics in humans.
For instance, in March 2016, RetroSense Therapeutics (Ann Arbor, Mich., USA) began the first-ever clinical safety trial of a novel optogenetics-based gene therapy called RST-001 to restore vision in blind patients with end-stage retinitis pigmentosa (RP). The technology involves injecting the retina with the light-sensing opsin channelrhodopsin-2 to create new sensitivity in retinal ganglion cells, an approach pioneered by Zhuo-Hua Pan of Ligon Research Center of Vision and Wayne State University in Ann Arbor. In September 2016, the global pharmaceutical and eye care giant Allergan (Dublin, Ireland) acquired RetroSense for US$60 million.
In January 2018, another optogenetics technology company, Paris-based GenSight Biologics, received approval from the U.K. Medicines and Healthcare Regulatory Agency (MHRA) to begin the PIONEER Phase I/II study of GS030, an injectable gene therapy for patients with end-stage RP. According to Bernard Gilly, CEO and cofounder of GenSight, the PIONEER study will be the first to study the safety and tolerability of an optogenetics-based gene therapy treatment for blindness in humans combined with a medical device.
 Blind patients suffering from retinitis pigmentosa may benefit from an optogenetic technique now in clinical trials. [GenSight Biologics]
Blind patients suffering from retinitis pigmentosa may benefit from an optogenetic technique now in clinical trials. [GenSight Biologics]
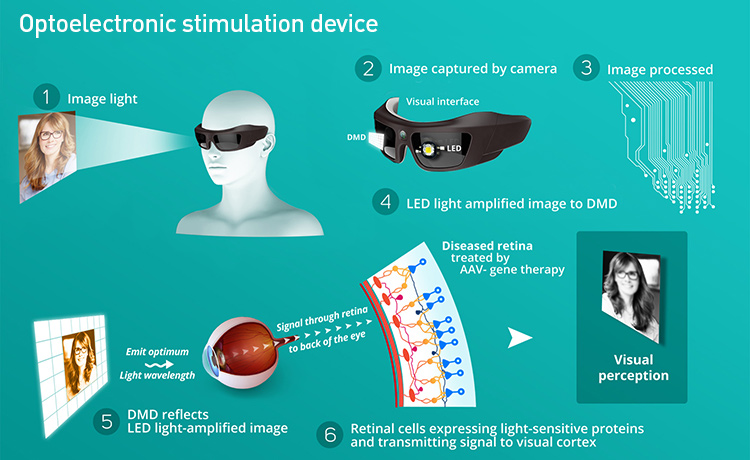
The GS030 approach starts with encapsulating a gene called ChrimsonR in adeno-associated virus (AAV), and injecting it into the eye, which is closed off to the body’s immune cells. Sensitive to red light at wavelengths between 590 and 630 nm, ChrimsonR is one of the various engineered opsins developed for safe use in animals by MIT’s Boyden. The virus will encode retinal ganglion cells with a boost of light-sensing capability, working alongside optoelectronic goggles worn by the patient to activate the cells. The goggles capture visual information using a camera, and convert it to a stream of safe, amplified red light sent to the enhanced retinal cells. The ganglion cells are stimulated, bypassing the degenerated photoreceptors and sending the captured visual scene to the healthy optical nerve and on to the brain. While these optogenetic approaches to RP are expected to restore only minimal vision in the blind, even the smallest gains could help patients recognize faces and navigate independently.
“It’s exciting to be involved in a project that could use optogenetics to restore sight in the blind,” said Gilly. “If proven in RP, our platform may also be able to help treat cochlear cell loss as well as conditions of the vagus nerve and the deep brain. We are hopeful that optogenetics is on the precipice of helping humans.”
Valerie C. Coffey is a freelance science and technology writer and editor based in Palm Desert, California.
References and Resources
-
E. Boyden et al. Nat. Neurosci. 8, 1263 (2005).
-
E. Boyden. F1000 Biol. Rep. 3, 11 (2011).
-
K. Montgomery et al. Nat. Methods 12, 969 (2015).
-
Galvan et al. J. Neurosci. 36, 3519 (2016).
-
Galvan et al. J. Neurosci. 37, 10894 (2017).
-
A. Shemesh et al. Nat. Neurosci. 20, 1796 (2017).
-
G. Shin et al. Neuron 93, 509 (2017).
-
L. Lu et al. Proc. Natl. Acad. Sci. USA, doi: 10.1073/pnas.1718721115 (2018).
-
J. Rogers. Curr. Opin. Neurobiol. 50, 42 (2018).
