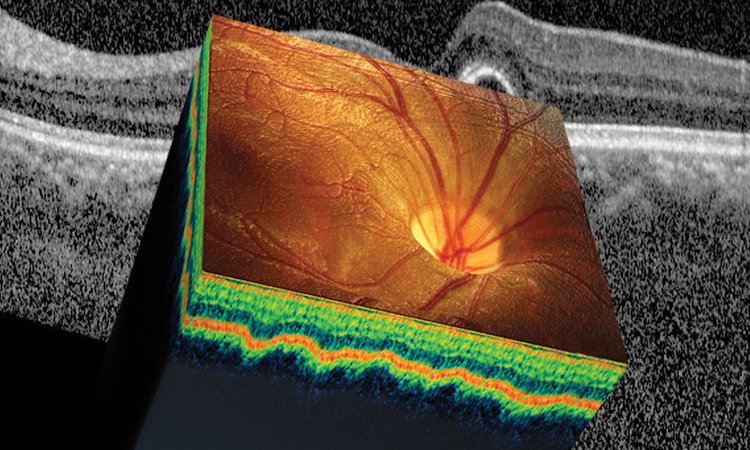
 Artist’s representation of an OCT cross section. [Illustration by Phil Saunders]
Artist’s representation of an OCT cross section. [Illustration by Phil Saunders]
This year marks the 25th anniversary of the celebrated initial optical coherence tomography (OCT) paper in Science by OSA Fellow James Fujimoto’s group at the Massachusetts Institute of Technology (Cambridge, Mass., USA). Five years later, in 1996, OCT profoundly changed the realm of ophthalmology when Carl Zeiss Meditec (Dublin, Calif., USA) launched the first commercial OCT platform for retinal imaging. In only 20 years, this non-invasive imaging technology has become a well-established fixture in the medical offices of ophthalmologists, gastrointestinal specialists, cardiologists and dermatologists. You might find OCT used to efficiently image through layers of tissue to detect early-stage skin cancer, view the placement of a stent in an artery, or help diagnose eye disease, all in real-time.
The firm Research and Markets (Dublin, Ireland) forecasts that the OCT market will continue to see a 12 percent compound annual growth rate through 2020 to reach US$1.32 billion. Recent developments behind this growth, such as supercontinuum sources, novel techniques and new swept-source systems, are reducing the cost and size of OCT platforms, and measuring new parameters in a multitude of new applications.
The firm Research and Markets forecasts that the OCT market will reach US$1.32 billion by 2020.
OCT is frequently compared to ultrasound, except that OCT uses light instead of acoustic waves. This enables OCT to measure small areas with higher resolution than ultrasound, depending on the wavelength of the source. The basis of OCT technology is an interferometry system that sends low-coherent light into both a sample and a reference arm of either a Michelson or Mach-Zehnder interferometer. The signal returns to a detector or line-scan camera, and the digitized signal is processed. The interference pattern is then used to create an image of the target. In OCT parlance, the resulting axial scan is called an A-scan. Multiple adjacent A-scans are then combined to create a B-scan, which is a cross-sectional image of the target.
The first OCT platform to reach commercialization in 1996 was based on time-domain OCT (TD-OCT), which used a moving reference mirror to mechanically capture light at rates of 400 A-scans/s. The mechanical nature of the scanning mirror provided good resolution for stationary subjects, but was limited in scan speed and image contrast.
The TD-OCT platform was followed in 2006 by the current popular iteration, spectral-domain OCT (SD-OCT). In SD-OCT platforms, a grating disperses multiple wavelengths of light into a detector array and then a processor, which performs a Fourier transform on the signal to construct the A-scan. Current commercial SD-OCT systems scan at rates up to 85 kHz, making them hundreds of times faster than the first TD-OCT systems. The increased scan-speed decreases the chances for motion artifacts, which means more efficient imaging and increased probability of identifying smaller features.
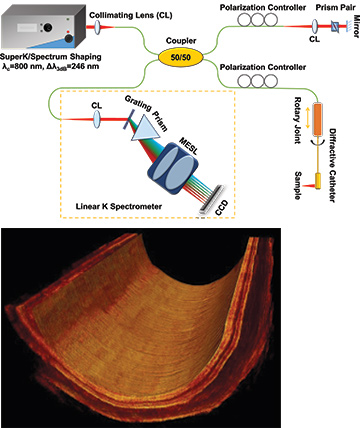 Xingde Li’s ultrahigh-resolution OCT (UHR-OCT) system uses a supercontinuum source from NKT Photonics (top)—and, in experiments on a guinea pig esophagus, achieved imaging quality (bottom) comparable to that of a system using a more expensive, bulky Ti:sapphire laser. [X. Li / Johns Hopkins University, Md., USA]
Xingde Li’s ultrahigh-resolution OCT (UHR-OCT) system uses a supercontinuum source from NKT Photonics (top)—and, in experiments on a guinea pig esophagus, achieved imaging quality (bottom) comparable to that of a system using a more expensive, bulky Ti:sapphire laser. [X. Li / Johns Hopkins University, Md., USA]
Commercial technology in research labs
Research laboratories serve as the first proving ground for commercial OCT platforms. In a recent Optics Letters paper, professor of biomedical engineering Xingde Li and colleagues Wu Yuan and Jessica Mavadia-Shukla at Johns Hopkins University (Baltimore, Md., USA) investigated how to optimize the operational conditions for an ultrahigh-resolution (UHR) OCT system in the spectral domain using a supercontinuum source for endoscopic imaging. A supercontinuum source enables a broader spectral bandwidth, which supports higher resolution imaging. In their work, Li’s team used a SuperK Extreme low-noise supercontinuum (SC) source, a compact tunable laser from NKT Photonics (Birkerød, Denmark). They were able to optimize the broad bandwidth (approximately 246 nm) around a central wavelength of 800 nm and use an appropriate power from the SC source to achieve shot-noise-limited operation in an SC-based UHR-OCT system. (See schematic and histology image below.)
“In OCT imaging, a commercial broadband source typically creates noise that causes poor image quality,” says Li. “We identified the optimal operational parameter space for the SuperK laser so that we can achieve competitive imaging performance in a much smaller package.”
Identifying an optimal reference-arm power of 6.1 µW and using an endoscope based on diffractive optics, the team obtained an axial resolution of 2.7 µm in air with a detection sensitivity of -107 dB. The group then successfully performed in vivo 3-D endoscopic imaging on a guinea pig esophagus, achieving 2,000 to 8,000 A-scans per frame at an imaging speed of up to 35 frames/s.
With this success, Li and colleagues found that the smaller SC source gives similar image quality to that of the bulky, expensive ultrafast Ti: sapphire laser traditionally used in laboratory-based UHR-OCT systems. In comparison, the new SC laser is turnkey, less expensive and doesn’t require a skilled operator. Another challenge in OCT endoscopic imaging is that moving the beam around from point to point requires a coupler that can handle the broader spectrum while rotating. The group plans to finesse a high-speed, broadband, fiber-optic rotary joint that would enable high-speed UHR-OCT endoscopic imaging in a more portable package.
“We look forward to moving this technology to commercialization for clinical study,” said Li. “We hope to enable early diagnosis of cancer and chronic obstructive pulmonary disease in luminal organs, and endoscopic UHR-OCT is well-suited to these in vivo applications.”
Such published studies from universities and research labs help set the stage for companies like NKT Photonics to develop the next commercial OCT platforms. But the path from exploratory work in university labs to commercialization is a long one.
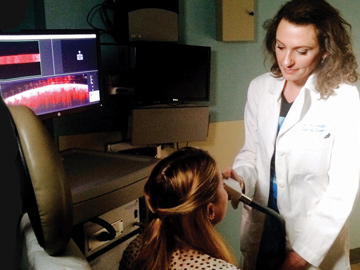 Dermatologist Orit Markowitz applies a hand-held VivoSight speckle-variance OCT imager from Michelson Diagnostics to identify vascular details that can help differentiate cancerous from non-cancerous lesions. [O. Markowitz / Mount Sinai School of Medicine, N.Y., USA]
Dermatologist Orit Markowitz applies a hand-held VivoSight speckle-variance OCT imager from Michelson Diagnostics to identify vascular details that can help differentiate cancerous from non-cancerous lesions. [O. Markowitz / Mount Sinai School of Medicine, N.Y., USA]
Clinical studies
Incorporating new tools to improve medical exams in clinics presents both monetary and time-commitment hurdles, so commercial entry into the OCT market often begins by partnering with major academic medical centers. Assistant professor and chief of dermatology Orit Markowitz at the Mount Sinai School of Medicine (New York, N.Y., USA) and colleagues used a VivoSight OCT scanner from Michelson Diagnostics (Maidstone, Kent, U.K.) in a pilot study to evaluate four early skin lesions that are difficult to differentiate. The real-time VivoSight—which has received European CE marking and regulatory clearance from the Australian Therapeutic Goods Administration and the U.S. Food and Drug Administration (FDA) 510(k)—enabled the team to visualize the specific mircrovascular patterns characteristic of each lesion to help identify them. Based on speckle-variance OCT (SV-OCT), the hand-held probe enables a dynamic en face (horizontal) scanning technique, whereas previous SV-OCT platforms used vertical sections.
Michelson’s SV-OCT platform uses a multi-beam, swept-source infrared (IR) laser with a center wavelength of 1305 nm to create, in seconds, 6×6-mm images from light scattering off moving red blood cells up to 2 mm below the skin’s surface. VivoSight achieves an axial resolution of 5 µm in tissue, with an A-scan rate of 20 kHz. A study found that VivoSight enabled proper identification of 48 percent more basal-cell carcinomas—the most frequently diagnosed type of cancer—than regular clinical exams. In addition to improved comfort for patients, proper identification helps to reduce the scarring and inconvenience of biopsy.
“What is exciting about the new speckle-variance feature is its potential to help better differentiate non-melanoma skin cancer,” said Markowitz. “It can also help differentiate between melanoma and dysplastic nevi.” Markowitz and her team are putting together a large “atlas” to help dermatologists differentiate the numerous types of pigmented lesions.
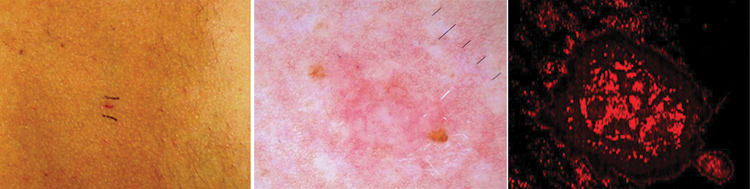 A comparison of clinical imaging techniques: (Left) Standard clinical imaging reveals a basal cell carcinoma (BCC) on a patient’s back. (Center) Dermoscopic imaging magnifies the lesion to reveal branching vessels characteristic of BCC. (Right) Speckle-variance OCT reveals more detail, including an array of thin, irregular vessels distributed focally throughout the BCC lesion and surrounded by normal vessels. [O. Markowitz / Mount Sinai School of Medicine, N.Y., USA]
A comparison of clinical imaging techniques: (Left) Standard clinical imaging reveals a basal cell carcinoma (BCC) on a patient’s back. (Center) Dermoscopic imaging magnifies the lesion to reveal branching vessels characteristic of BCC. (Right) Speckle-variance OCT reveals more detail, including an array of thin, irregular vessels distributed focally throughout the BCC lesion and surrounded by normal vessels. [O. Markowitz / Mount Sinai School of Medicine, N.Y., USA]
Going wide and deep
The biggest markets for OCT are ophthalmology, oncology and angiography (the imaging of blood vessels). In ophthalmology—the primary market—it’s critical to maximize the scan-speed to prevent eye-motion artifacts without sacrificing image quality. To this end, Heidelberg Engineering (Heidelberg, Germany) integrated SD-OCT with confocal scanning-laser ophthalmoscopy (cSLO) into an OCT platform called SPECTRALIS. The cSLO technique uses focused laser-light imaged through a confocal pinhole to provide a sharp, high-contrast image. The addition of cSLO to SD-OCT improves image quality and is more comfortable for patients while enabling high scan rates.
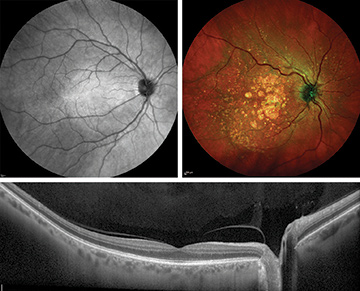 Top: The SPECTRALIS spectral-domain OCT platform with widefield-imaging module reveals a widefield image of the fundus (back portion) of a healthy eye (upper left in IR), with the macula in the center and optic disc toward the right. An image in multicolor-scanning mode (upper right) shows geographic atrophy and drusen. Bottom: The OCT line scan reveals a cross-section of a healthy retina, including the optic nerve head (pit on the right). [Heidelberg Engineering, Germany]
Top: The SPECTRALIS spectral-domain OCT platform with widefield-imaging module reveals a widefield image of the fundus (back portion) of a healthy eye (upper left in IR), with the macula in the center and optic disc toward the right. An image in multicolor-scanning mode (upper right) shows geographic atrophy and drusen. Bottom: The OCT line scan reveals a cross-section of a healthy retina, including the optic nerve head (pit on the right). [Heidelberg Engineering, Germany]
SPECTRALIS has five lasers: blue at 486 nm, green at 518 nm, near-IR at 786 nm, far-IR at 815 nm and “superluminous” at 870 nm. It can scan the retina in a couple of seconds, depending on the selected scan pattern, to simultaneously acquire combinations of several imaging modes, such as fluorescein angiography and OCT, or multicolor scanning laser and OCT. To further increase precision and reliability in the SPECTRALIS platform, Heidelberg introduced “TruTrack” active eye tracking, which simultaneously images with two beams of light, one of which maps over 1,000 points in real time to reference eye movement and mitigate eye-motion artifacts.
A technology called OCT angiography, based on standard OCT scans, can image the microvasculature system of the retina and choroid (a vascular layer of the eye beneath the retina) to help study vascular abnormalities. To acquire sufficiently dense volume scans in a relatively short time, acquisition speed plays a pivotal role in OCT angiography. Heidelberg Engineering recently launched a module called OCT2 with a scan rate of 85 kHz, which will enable an upcoming OCT angiography platform. (The platform is not yet for sale; no launch date has been announced.)
A typical SD-OCT will image a field of about 20 × 20 degrees. However, eye diseases such as diabetic retinopathy require a much wider field of view (FOV). So it’s also critical to maximize the FOV without sacrificing resolution or scan speed. Heidelberg’s new widefield-imaging module adds functionality to the SPECTRALIS SD-OCT platform with a 55-degree FOV, providing multiple perspectives in the retina, which the company says can simplify diagnostic protocols and help in the detection of peripheral pathology.
A new wave in the OCT market
SD-OCT is a solid technology with room for market growth, but it has its challenges. The SD-OCT platform uses a wide wavelength band to reflect off the different layers in the eye. The deeper parts of the eye are imaged with the higher-frequency interference fringes, which are more difficult to detect, producing a lower-signal-to-noise image of the deeper structures.
In the past year or two, according to Nate Kemp, market development manager at Axsun Technologies (Billerica, Mass., USA), another OCT technology has begun to make waves, shifting the attention away from SD-OCT: swept-source OCT (SS-OCT). Companies are in a race to launch SS-OCT platforms that can create high-resolution images that are deeper, wider and more detailed than previous OCT platforms.
Companies are in a race to launch SS-OCT platforms that can create high-resolution images that are deeper, wider and more detailed than previous OCT platforms.
In comparison to SD-OCT, SS-OCT uses a tunable laser to rapidly scan at rates of up to 100,000 A-scans/s sequentially through a range of wavelengths to assemble the B-scan cross-sectional image, often in a fraction of a second. The optic nerve head, sclera, vitreous and choroid structures can be imaged all in one fast, detailed scan.
While SD-OCT systems are typically based on a central laser wavelength of 840 or 850 nm, swept-source tunable laser systems are centered around 1 µm (for example, at 1030, 1050 or 1060 nm), which penetrates tissue more deeply than 850 nm. The longer wavelengths can penetrate opaque media such as cataracts more effectively, and provide the most accurate biometry measurements possible for the best selection of an intraocular lens (IOL) before cataract surgery.
This latest iteration of OCT imaging has arrived in a few guises. First was Topcon in 2012 with a commercial retinal swept-source OCT (available in select countries), the DRI OCT Atlantis, with 100,000 A-scans/s at 1050-nm central wavelength. Then, for several years, competitive development of other SS-OCT platforms progressed in relative stealth-mode, while clinical trials endeavored to evaluate the new platforms and move the needle to see what would catch on.
In September 2015, Heidelberg previewed a new SS-OCT platform: the cataract and refractive imaging platform. The system is still under development, but when available, it will have options for biometry, corneal topography/tomography and anterior imaging functions.
“Swept-source technology allows doctors to see what they measure and measure what they see,” said Kester Nahen, managing director at Heidelberg Engineering. “That will be a very distinctive feature.” But timelines for necessary regulatory clearances remain uncertain.
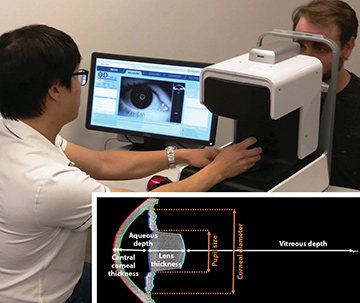 An ophthalmologist uses the ARGOS advanced optical biometer to measure a patient’s eye parameters before cataract surgery. (Inset) One ARGOS scan can obtain six simultaneous measurements of different parts of the eye in 0.6 seconds. [Santec / Movu, Calif., USA]
An ophthalmologist uses the ARGOS advanced optical biometer to measure a patient’s eye parameters before cataract surgery. (Inset) One ARGOS scan can obtain six simultaneous measurements of different parts of the eye in 0.6 seconds. [Santec / Movu, Calif., USA]
In October 2015, laser and optical components manufacturer Santec (Aichi, Japan) received FDA 510(k) approval to market ARGOS, the advanced optical biometer to measure eye parameters of patients before cataract surgery. ARGOS uses Santec’s high-performance, high-speed swept-source laser centered at 1060 nm to enable real-time 2-D imaging of the eye from cornea to retina at high resolution of approximately 10 µm standard deviation for axial length. To measure corneal curvature, the SS-OCT system uses a reflected image from a ring of 16 IR LEDs.
In one 0.6-s measurement, the ARGOS biometer obtains six images, measuring three sets of nine essential parameters for IOL selection: axial length, corneal thickness, anterior chamber depth, lens thickness, pupil size, corneal diameter, K-values (K1, K2) and toric angle. The IOL power is calculated from one of several power-formula choices. In clinical trials, ARGOS demonstrated repeatability and reproducibility of measurements similar to existing biometers, but with improved acquisition rates for axial length in dense cataracts.
Santec has created the new company Movu (Santa Clara, Calif., USA) to introduce ARGOS to clinical markets.
“The FDA approval to market the ARGOS biometer is a major milestone, one that we hope will help the ophthalmic clinician improve effective surgical outcomes for cataract patients,” said Jonathan Evans, vice president of sales at Santec.
In May 2016, Carl Zeiss Meditec (Jena, Germany) received the CE mark of approval for the PLEX Elite 9000, an SS-OCT platform introduced for clinical research use among the Advanced Retina Imaging Network, a collaboration with leading clinicians and researchers around the globe. The Advanced Retina Imaging Network began performing clinical research in patient care in July 2016. The consortium will share feedback that will help determine the direction of new clinical applications and future commercial offerings.
Similar to other SS-OCT offerings, Zeiss’ platform for OCT and OCT angiography has a swept-source tunable laser centered at 1050 nm, and a scan speed of 100,000 A-scans/s at a tissue depth of 3.0 mm. The axial resolution is 6.3 µm, with a 56-degree FOV. The PLEX Elite 9000 is available in Europe, the United States and selected other countries for investigational use only, pending 510(k) clearance with the FDA.
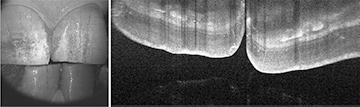 A line scan (right) taken with a briefcase-sized swept-source OCT platform enabled by Axsun’s swept laser integrated engine reveals a cross-sectional image of teeth with details in the enamel and dentin layers. [Nate Kemp / Axsun Technologies, Mass., USA]
A line scan (right) taken with a briefcase-sized swept-source OCT platform enabled by Axsun’s swept laser integrated engine reveals a cross-sectional image of teeth with details in the enamel and dentin layers. [Nate Kemp / Axsun Technologies, Mass., USA]
The elephant in the room
In addition to new SS-OCT products in the development stage, companies continue to create experimental platforms and gather clinical data to clear regulatory hurdles. At the sub-component level, manufacturers like Axsun are developing lower-cost pieces like the tunable laser and integrated engine hardware so that end users and systems integrators can build SS-OCT platforms that are lighter and consume less power.
“Because the cost of OCT hardware has been so high,” says Kemp, “it can be hard to get commercial traction. Ophthalmology has a large installed base that enables a reasonable profit margin. In cardiology, the profit comes from the annual sale of thousands of disposable catheters that go along with the OCT system. But in other markets, the cost must be low at the component level to reduce the initial cost barrier.” Axsun decreases the cost by integrating very small, lower-power laser beams with Ethernet-based data acquisition electronics.
Another shifting paradigm is the size of OCT platforms. The typical large, bulky, roll-around console is expensive in terms of infrastructure and workflow complexity needed to obtain an OCT image, according to Kemp.
“The system has to be rolled in, plugged in, booted up, and it takes up a large area in the office,” said Kemp. Incorporating their more integrated, low-power, laptop- and tablet-ready laser beam component, Axsun demonstrated a one-off SS-OCT system at the Photonics West 2016 conference in February that is briefcase-sized, battery-powered and pointing to the future. The demo featured a 100-kHz swept laser (customizable to either 1060 or 1310 nm), a hand-held probe, and scan rates of more than 125 frames/s.
Just as new types of ultrasound equipment are becoming more shoebox-sized with the same level of image quality, smaller OCT platforms seemed destined to follow.
“It doesn’t have to be the elephant in the room anymore,” said Kemp.
Valerie C. Coffey is a freelance science and technology writer and editor roving the United States in a 45-foot motorcoach with a mobile hotspot.
References and Resources
-
R.F. Spaide et al. “What’s next in laser and OCT?” Rev. Opthal. (March 2013).
-
J. Kolb et al. “Ultra-widefield retinal MHz-OCT imaging with up to 100 degrees viewing angle,” Biomed. Opt. Express 6, 1534 (2015).
-
O. Markowitz et al. “Speckle-variance optical coherence tomography: A novel approach to skin cancer characterization using vascular patterns,” Dermatol. Online J. 22, 4 (2016).
-
W. Yuan et al. “Optimal operational conditions for supercontinuum-based ultrahigh-resolution endoscopic OCT imaging,” Opt. Lett. 41, 250 (2016).
