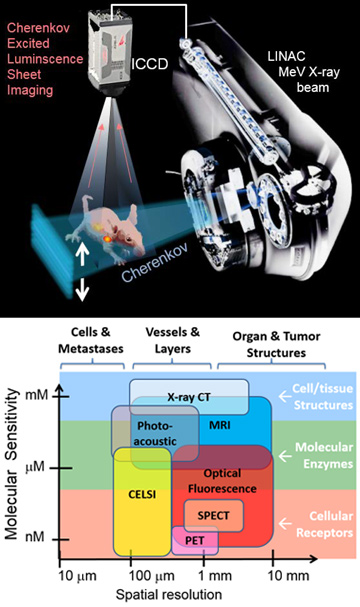
 (Top) CELSI imaging uses a linear accelerator (LINAC) beam, shaped to sheet-illuminate a rodent with Cherenkov radiation. Gated luminescence from the animal is imaged from above using an intensified CCD (ICCD) camera. (Bottom) The imaging approach fills a niche in the molecular-imaging world, with the highest spatial resolution (100–300 microns) and molecular sensitivity (micromolar to nanomolar), and works for biologically compatible organic molecules.
(Top) CELSI imaging uses a linear accelerator (LINAC) beam, shaped to sheet-illuminate a rodent with Cherenkov radiation. Gated luminescence from the animal is imaged from above using an intensified CCD (ICCD) camera. (Bottom) The imaging approach fills a niche in the molecular-imaging world, with the highest spatial resolution (100–300 microns) and molecular sensitivity (micromolar to nanomolar), and works for biologically compatible organic molecules.
A fundamental limitation in all visible-light molecular-imaging systems has been the strong scattering of light in biological tissues, which has limited molecular imaging to superficially thin tissue slices, exposed surfaces, or diffuse, blurry images of whole animals. This year, our group outlined a process for using thin sheets of ionizing radiation to induce optical Cherenkov light excitation in thick scattering media, converting the thick-tissue imaging problem into a linear inversion that provides high-resolution localization of molecular signals from inside the animal.1 We believe that the realization that MeV ionizing radiation directly transmits through tissue, and can be used to optically illuminate the tissue from within, could prove a landmark step towards high-resolution molecular imaging.
The approach, called Cherenkov-excited-luminescence sheet imaging (CELSI), is analogous to light-sheet microscopy, which has changed the capabilities of fluorescence microscopy, allowing full-field imaging and reducing scatter and out-of-plane background signals. CELSI whole-body imaging through an entire rat body was first demonstrated by our group in 2015.2 Our work this year has shown that using the light-sheet geometry of orthogonal excitation–emission lets us locate the source by applying the depth-dependent attenuation correction and deconvolution methods that are standard in imaging systems such as X-ray, nuclear medicine and ultrasound.
With the source localized at high resolution in this way, the image can be recovered using linear corrections and mathematics, instead of unstable nonlinear approaches involving diffusion theory—thereby avoiding the limitations of dense optical scatter that cause deep regions to be poorly resolved by conventional optical imaging. Using CELSI, we have demonstrated molecular-imaging sensitivity in the micromolar to nanomolar range, essential for in vivo metabolic and immunologic imaging.3 CELSI can be used for all classes of biologically compatible organic molecules used in traditional in vivo molecular imaging.
Perhaps most exciting, the technology to produce precise, thin sheets of radiation is widely available in radiation oncology centers, where multi-leaf collimators on the outputs of linear accelerators have advanced to a state at which dynamic three-axis shaped-beam treatments are standard care. CELSI’s spatial resolution is limited only by the collimator’s step size of 100 microns. This technology—which affords high-resolution molecular imaging deep in mammalian tissue, with sub-micromolar sensitivity—thus opens up horizons for in vivo molecular imaging in rodents today and, potentially, for human cancer patients tomorrow.
Researcher
Brian W. Pogue, Dartmouth University, N.H., USA
References
1. P. Bruza et al. Opt. Lett. 41, 2986 (2016).
2. R. Zhang et al. Opt. Lett. 40, 827 (2015).
3. B.W. Pogue. Opt. Photon. News 26(9), 24 (2015).
