Feature
Multimodal Nonlinear Optical Microscopy: From Biophotonics to Geophotonics
Nonlinear optical spectroscopies such as CARS and SRS offer rapid, label-free, chemical-specific imaging. And the ability to simultaneously deploy multiple modes of nonlinear imaging on a single sample is creating some striking new views of natural materials.
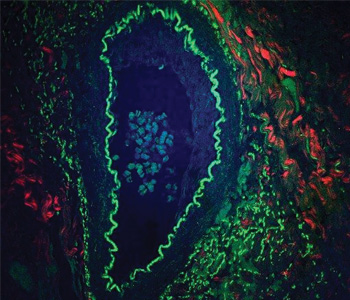 Application: Biophotonics. Multimodal CARS image of an unstained rabbit arteriole. A 50-μm thick section of arteriole, tangentially cut to the lumenal side, reveals lipids (red zones, CARS), collagen (blue zones, SHG) and smooth muscle elastin (green zones, TPEF). All three channels were obtained simultaneously from a single sample.
Application: Biophotonics. Multimodal CARS image of an unstained rabbit arteriole. A 50-μm thick section of arteriole, tangentially cut to the lumenal side, reveals lipids (red zones, CARS), collagen (blue zones, SHG) and smooth muscle elastin (green zones, TPEF). All three channels were obtained simultaneously from a single sample.
Linear optical microscopy techniques, such as transmission or laser fluorescence microscopy, are ubiquitous in medical research and diagnostics. And, since nature did not color-code biological materials for our viewing pleasure, these methods typically rely on exogenous stains or fluorophores, such as organic dyes and quantum dots, to provide high contrast when imaging with visible light.
…Log in or become a member to view the full text of this article.
This article may be available for purchase via the search at Optica Publishing Group.
Optica Members get the full text of Optics & Photonics News, plus a variety of other member benefits.
