 All photos in this article were taken by Erin Monacelli. Specimens are from the Natural History Museum of Los Angeles.
All photos in this article were taken by Erin Monacelli. Specimens are from the Natural History Museum of Los Angeles.
As any optical engineer knows, the fabrication of optical filters is no simple task. Such filters and reflectors are produced by vacuum deposition, and they require an array of sophisticated equipment: a high-vacuum chamber, very pure deposition materials, a means for sputtering or evaporating these materials, thermal control and a mechanism for monitoring the thicknesses of the materials deposited.
Mother Nature, on the other hand, pulls off spectacular filters without any help. Butterflies, moths, beetles, birds and fish self-assemble filters that are just as diverse, complex and visually striking as humankind’s best efforts.
Interference effects
Interference effects occur when two optical waves constructively or destructively combine after reflecting from two parallel surfaces. If the optical path of the two surfaces is a multiple of exactly one full wavelength, the two waves will constructively interfere and there will be a strong reflection at that wavelength.
If, on the other hand, the phase of the reflected wave differs by one-half wavelength or an odd multiple of one-half wavelength, then the reflected waves will be completely out of phase; destructive interference will occur at that wavelength. In the latter case, the reflected wave will be weak, and therefore maximum in transmission. The phase of the two waves depends on the thickness and refractive index of the media separating the two surfaces, the wavelength of the light and angle at which light strikes the surfaces.
 Emerald swallowtail butterfly (Papilio palinurus, Linnaeus, 1758)
Emerald swallowtail butterfly (Papilio palinurus, Linnaeus, 1758)
 Urania swallowtail moth (Urania fulgens, Walker, 1854)
Urania swallowtail moth (Urania fulgens, Walker, 1854)
 Madagascan sunset moth (Chrysiridia rhipheus, Hübner, 1823)
Madagascan sunset moth (Chrysiridia rhipheus, Hübner, 1823)
Constructive or destructive interference will be the most intense and purest when waves reflected from both surfaces have the same amplitude. If instead of one thin film or surface, multiple parallel surfaces exist of the correct thickness, refractive index and incident angle, then even more intense interference colors can be obtained. If incident light is comprised of a full spectrum of “white” light, only one color will satisfy the conditions for constructive interference. However, when incident light is monochromatic, the light interference pattern will consist of a series of bright and dark bands.
Interference effects: moths and butterflies
The emerald swallowtail butterfly (Papilio palinurus, Linnaeus, 1758), which is native to Indonesia, exhibits interesting properties of both additive color and interference effects. The wing scales of this butterfly show that its surfaces are comprised of a regular two-dimensional array of concave depressions that are 4 to 6 µm in diameter and 0.5 to 3 µm deep. The depressions consist of approximately ten multi-layer index pairs that reflect yellow light from the depression bottoms and retro-reflect blue light from the 45-degree sides.
Since the structures are too small to be resolved by eye, the colors are additive. In other words, just like the colors we see on our television screen, red, green and blue dots are used to create an entire color palette.
Moths are usually thought of as having dull colors such as brown and gray. There are exceptions, however—notably the Urania swallowtail moth (Urania fulgens, Walker, 1854), which can be found in the U.S. state of Texas and the continent of South America, or the sunset moth of Madagascar (Chrysiridia rhipheus, Hübner, 1823).
Like the swallowtail butterfly, these moths are iridescent. Each scale is comprised of four or five layers of cuticle, approximately 40 nm thick. These cuticle layers are held apart by tiny cuticle rods that allow air pockets of approximately 100 nm to form a Fabry-Perot filter. By altering the spacing and thickness of the cuticle layers and incorporating different colored pigments within the scale, the moths create various colors.
The Morpho butterfly (Papilio achilles, Linnaeus, 1758) is another striking example of brilliant iridescence produced by interference. In this case, the color is not created by a series of high- and low-index layer pairs, but rather through tree-like structures or lamellae that are self-assembled from cuticles that have an index of refraction of 1.56.
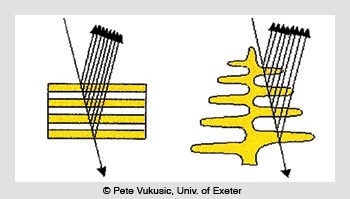 Iridescent reflection from cuticles (left) and lamellae (right)
Iridescent reflection from cuticles (left) and lamellae (right)
 Morpho butterfly (Papilio achilles, Linnaeus, 1758)
Morpho butterfly (Papilio achilles, Linnaeus, 1758)
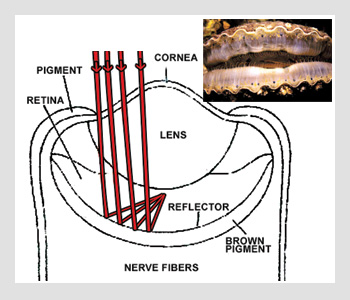 Scallop eye
Scallop eye
There are six to ten branches on each side of the lamellae trunk (depending on the Morpho species), with a density of 700 to 2,000 lamellae per mm. These self-assembled multi-cavity Fabry-Perot structures create beautiful iridescent blue colors with reflectance values from 45 to 65 percent at peak wavelengths ranging from 400 to 460 nm, with 150- to 300-nm-wide spectral bands, depending on the species.
Mirror optics: the scallop eye and other sea invertebrates
In the world of optical engineering, reflectors can be produced by evaporating common metals such as aluminum or gold, or by fabricating reflectors composed of constructively interfering stacks. Scientists believe that the function of reflectors in the eyes of many insects and animals is to provide a double pass through the retina so that it can absorb as many photons as possible.
Some modern detectors use this same principal to gain quantum efficiency. In the eyes of animals, this improves detection in low light conditions. Reflectors called tapetum lucidum (Latin for “bright tapestry”) are primarily found in nocturnal animals or those that live in low-light environments, such as scallops, bottle nose dolphins, sharks, alligators, cats, deer and bush babies.
A tapetum is a reflecting, self-assembled multi-layer located immediately behind (and sometimes within) the retina. It reflects unabsorbed photons back through the retina for another chance at absorption. The tapetum is not present in the human eye, which is why humans have such poor night vision. In people, unabsorbed photons can be seen when we observe the “red eye” effect in photos. The redness does not represent light reflected from the retina but rather due to light reflecting off the back of the eye, through the blood-rich (and non-reflective) choroid. In animals, reflection from the tapetum frequently appears to be glowing in one of a variety of colors, including blue, green, yellow and pink.
Man-made telescopes consist of lenses and metallized mirrors in multiple mirror configurations. There are also animal eyes that look a lot like a man-made catadioptric optical system design, incorporating both a refractive lens and reflector for folding the light path. The optical configuration shown in the bottom figure on the right is that of a scallop eye.
About 50 to 100 of these eyes can be found around the top and bottom of a typical scallop shell. Each eye is a miniature optical system measuring approximately 1 mm in diameter. The aspheric cornea and lens bend rays, and a reflector folds the image back on itself, where photons are detected. Several layer pairs of guanine (n=1.83) and cytoplasm (n=1.33) make up the concave eye reflector. Each layer is approximately 100 nm thick with an equivalent optical thickness of one-quarter wave between 525 and 575 nm, the transmission maximum of sea water. With ten layer pairs, the constructive interference of this reflector is close to 100 percent.
Photonic band gaps: peacock feathers and opals
 Male peacock feathers
Male peacock feathers
Peacock feathers and opals share an interesting configuration that makes use of photonic band gap structures. By carefully examining peacock feathers, one will see a central stem that has barbs projected from the side of it. Off the sides of the barbs are thousands of flat barbules, which, in the male peacock, are responsible for the blue, green, yellow and brown iridescent colors.
A transverse cut through a barbule shows a 2D photonic structure composed of self-assembled melanin rods connected by keratin and surrounded by a square array of air spaces. The lattice constant or rod spacing of melanin is 140 nm, 150 nm and 165 nm to produce the blue, green and yellow colors, respectively. To obtain a saturated color, 3D structures are required. In the peacock feather, the number of periods or rod layers in the structure is increased—nine to twelve for blue and six for yellow.
A 3D periodic structure also occurs in opals. Electron images of the opal show that it is made up of 3D matrix of silicate spheres about 150 to 300 nm in diameter. The beautiful play of colors is produced by interference from this three-dimensional repetitive structure. When illuminated, they reflect light and act as a three-dimensional array of coherent point sources. When an opal is rotated, the effective spacing between the silicate spheres changes, thus altering the colors seen. The size over which the array maintains the same periodicity determines the color. Large uniform areas are rare.
 Japanese metallic wood-boring beetle (Chrysochroa fulgidissima, Schönherr, 1817)
Japanese metallic wood-boring beetle (Chrysochroa fulgidissima, Schönherr, 1817)
 Jewel scarab (Chrysina optima, Kirby, 1828)
Jewel scarab (Chrysina optima, Kirby, 1828)
 Frog-legged leaf beetle (Chrysomelidae sagra femorata, Drury, 1773)
Frog-legged leaf beetle (Chrysomelidae sagra femorata, Drury, 1773)
Multi-layer reflectors: beetles and fish
Researchers have replicated the many colors of the wood-boring beetle by performing computer simulations of a multi-layer structure consisting of a combination of air (70 nm), keratin (50 nm) and melanin (170 nm); this provided a fairly accurate color rendition of the actual beetle that is shown in the bottom photograph on the left.
While many beetles are green, that is not the only color that they display. Beetles come in a variety of colors, including white, black, brown, blue and gold, as seen in the jewel scarab or the frog-legged leaf beetle. In some instances, color is produced by a regular fixed spatial periodicity of multilayer stacks that consists of high- and low-index materials that are sometimes over-coated with a scattering structure.
Broadband reflectors can be constructed by more than one method. One approach is to self-assemble multiple reflecting stacks (one for each color), and then arrange these reflecting stacks on top of one another. Another way is to systematically change the optical thickness of the component layers to reflect the range of desired colors. This so-called “chirped” reflecting stack is what gives some gold beetles their brilliant color.
Whether the sky is sunny or cloudy, the illumination under water is almost constant in a horizontal plane. The intensity does, however, decrease rapidly with increasing depth. This fact alone is useful in camouflage. Since fish scales are broadband high-reflectors, they always reflect light to an observer with the same intensity that would be seen even if the fish were not present.
But what if a predator is below its prey looking upwards against the bright down-welling light? In the ocean, many animals can produce light; often they can direct this light in a particular direction by using a type of mirror. The light produced in photophores is used to fill in the dark obscuration created by fish that are between the sun and the predator.
In the deep sea hatchet fish, photophores are arranged in groups. Light is produced in a central chamber that feeds many light tubes in the group. In this deep-dwelling fish, these light tubes, with their different intensities and light properties, shine downwards and sideways to provide the camouflage needed for the ocean depths.
Summary
Many of natures’ creatures biologically self-assemble a variety of optical filters and reflectors that produce an outstanding multiplicity of effects without the use of the sophisticated equipment required by humans. The filters produced can be used for communications among like species, for mating, camouflage, to increase detection capabilities, to startle or confuse a predator, for illumination, and for many other applications.
The filters and reflectors produced typically function either as multi-layer interference structures or by photonic band gap principles. These varied and sophisticated filters and reflectors are naturally produced (and at low temperatures!) with precision that we can only hope to learn to reproduce.
The author would like to acknowledge the gracious assistance of Weiping Xie, Collections Manager of the Entomology Department at the Natural History Museum of Los Angeles County for providing the specimens photographed in this article by Erin Monacelli.
H.D. Wolpert is director of engineering at Bio-Optics in Los Angeles, Calif., U.S.A.
References and Resources
>> M.F. Land “The optics of invertebrate eyes,” In: Tutorials in Optics, editor E.D. Moore, 1-14, Washington, D.C., Optical Society of America (1992).
>> P. Vokusic et al. “Structural color: color mixing in wing scales of a butterfly,” Nature 404, 457 (2000).
>> S. Kinoshita et al. “Photophysics of structural color in the morpho butterflies,” Forma 17, 103-21 (2002).
>> J. Zi et al. “Coloration strategies in peacock feathers,” Proceedings of the National Academy of Sciences of the U.S., October 2003.
>> D. Falk et al. Seeing the Light; Optics in Nature, Photography, Color, Vision and Holography, John Wiley & Sons, N.Y., 1986.
>> H. Hirayama et. al. “Rendering iridescent appearing on natural objects,” Eighth Pacific Conference on Computer Graphics and Applications (PG’’00), 2000.
