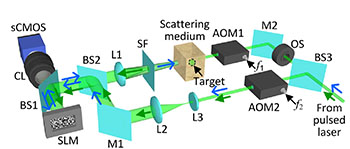
Schematic of the focusing setup for b-TRAP. AOM, acousto-optic modulator; BS, beam splitter; CL, camera lens; L, lens; M, mirror; OS, optical shutter; sCMOS, scientific complementary metal oxide semiconductor camera; SF, spatial filter (zero-order block); SLM, spatial light modulator. [Image: Ma et al., Optica, doi: 10.1364/OPTICA.2.000869]
Researchers continue to search for solutions to the problem of focusing light in scattering media—an essential requirement for optical imaging of biological tissues at any meaningful depth. A team of scientists from Washington University of St. Louis (USA) now reports that it has devised an improvement to wavefront-shaping techniques that can substantially boost the speed, quality, and practicality of imaging in scattering media (Optica, doi: 10.1364/OPTICA.2.000869). The team sees applications for the technique ranging from deep-tissue biophotonic imaging to real-time capture of movement and flow of blood cells in living tissue.
The need for speed
The new technique builds on an approach reported in 2014 by the same researchers. That technique, called time-reversed adapted-perterbation optical focusing (TRAP), uses successive laser shots to illuminate the targeted tissue at different time steps. The signals are then subtracted, and the difference between them—attributable to motion or dynamics within the tissue—is used as a “virtual guide star” for focusing the image. Specifically, the optical wavefront from the guide star is time-reversed using digital optical phase conjugation, to cancel out the effects of scattering from the static part of the tissue and allow for a focused image of the moving target.
While the principle of TRAP is simple, the technique has had an Achilles’ heel: speed. As a practical matter, the technique has required four laser shots, synchronized with four separate camera exposures, to complete a single field measurement. That has held the shortest practical time between field measurements to around 60 ms—too long to capture many biological processes.
A single-exposure method
To boost TRAP’s capabilities to capture faster processes, the Washington University team, led by OSA Fellow Lihong Wang, made two innovations. First, they devised a setup where the perturbed optical field could be captured in a single camera exposure, on a timescale defined by the 6-ns width of the laser pulse (rather than the longer timescale of multiple camera exposures). This, according to the team, shrinks the single-measurement time by seven orders of magnitude, to 6 ns rather than 60 ms. And, second, the new approach encodes the intensity holograms used in the optical phase conjugation step in a binary scale—with areas exceeding the mean intensity encoded as 1 and areas beneath that threshold encoded zero—rather than using much more processor-intensive grayscale values.
According to the team, the modified approach, labeled binary TRAP or b-TRAP, allows the field measurements to be taken and processed at short enough intervals to allow tracking of moving objects and flow, such as blood flow, within scattering media—at investigation depths and timescales relevant to biological systems. The researchers conclude that the technique “has anticipated applications in deep-tissue molecular imaging, flow measurement and cytometry, photodynamic therapy, optogenetics, microsurgery, and more.”
